糖尿病并发症(专业版)
糖尿病是以慢性高血糖为特征的终身代谢性疾病。长期高血糖形成的糖毒性,导致神经和血管损伤病变,累及全身性组织和器官,产生多种并发症而危及糖尿病患者的生命安全。
其他名称:糖尿病合并症
英文名称:Diabetes Complications
1.糖尿病急性并发症:包括糖尿病酮症酸中毒、乳酸性中毒和高血糖高渗性昏迷等。发病原因主要是由于胰岛素重度缺乏及升糖激素异常升高,导致血糖过高,引起糖、脂肪和蛋白质代谢紊乱,以致机体水、电解质和酸碱平衡失调等急性发作。此外,在糖尿病降糖治疗过程中突然出现的低血糖昏迷反应。
2.糖尿病慢性并发症:这是糖尿病致残、致死的主要原因,主要类型如下:
在稳定健康的血糖水平上,下列干预路径和措施和有助于抑制糖尿病并发症发生和发展。
1.抑制糖基化终产物(AGEs):
糖基化是葡萄糖和其他糖与蛋白质、脂质和核酸不可逆地结合,导致它们功能失调的过程。糖基化产生的功能失调分子被称为晚期糖基化终产物(AGEs)1,2,3。AGEs通过抑制神经功能(进而影响其活动)和触发炎症反应(更多损害神经细胞)来损伤神经4。
1.1.苯磷硫胺:
糖尿病和肥胖通常会引起硫胺素(维生素B1)相对缺乏,这会导致高血糖的破坏性作用5,6。苯磷硫胺是硫胺素(维生素B1)的脂溶性衍生物,其生物利用度比硫胺素高得多,并且能够在血液中达到口服硫胺素浓度的数倍7。这种独特形式的维生素B1可抑制AGEs的形成、炎症和氧化应8,9,6。
一项针对165名糖尿病神经病变患者的临床试验发现,连续六周补充苯磷硫胺可以减轻糖尿病神经病变的疼痛。与每天服用300mg的受试者相比,每天服用600mg苯磷硫胺的益处更为明显。此外,服用苯磷硫胺的受试者益处更长久10,11。
在一项针对13名2型糖尿病受试者的临床试验中,参与者在每天1050mg的苯磷硫胺3天疗程前后吃了一顿高AGEs餐,在两次高AGEs餐后评估受试者的血管和内皮功能。苯磷硫胺给药完全预防了血管功能障碍的迹象,内皮功能障碍和氧化应激的生物标志物显著减少12。
临床和动物研究已经证明了苯磷硫胺在治疗糖尿病相关神经病变、肾病、外周血管疾病和视网膜病变方面的疗效13-15。
1.2.肌肽:
肌肽能够抑制AGEs的形成,甚至逆转蛋白质糖基化16。在一项针对糖尿病小鼠的研究中,补充肌肽使血浆肌肽水平增加了20倍,甘油三酯水平降低了23%,并提高了动脉粥样硬化病变的稳定性17。肌肽还被证明可以提高细胞在高浓度葡萄糖存在下的生存能力,并改善糖尿病动物的伤口愈合18。糖尿病动物模型显示,补充肌肽可以提高红细胞改变其形状的能力,这是血液流动过程中遇到的机械力所必需的;这一过程在糖尿病中受损,导致糖尿病并发症19。
1.3.维生素B6(吡哆醛):
维生素B6,特别其活性形式5磷酸吡哆醛(PLP,简称吡哆醛)参与葡萄糖代谢的几个方面,是一种有效的抗糖化剂20。除了防止蛋白质糖基化,PLP还是脂质(脂肪)糖基化的有效抑制剂21。与健康对照组相比,糖尿病患者的脂质AGEs升高,脂质AGEs的积累可能导致与糖尿病相关的血管疾病22。AGEs的高膳食摄入也会增加糖尿病患者的心血管疾病风险。
较低的血清PLP水平与糖尿病的发病率和进展有关,维生素B6的抗糖化特性可能有助于预防糖尿病并发症20。研究表明,在日本2型糖尿病患者中,维生素B6的摄入量越高,糖尿病视网膜病变的发生率越低23。
对人类和动物的干预研究也很有前景。例如,用35mg PLP、3mg活性叶酸和2mg维生素B12治疗20例2型糖尿病患者,可改善糖尿病周围神经病变的皮肤感觉24。在一项随机对照试验中,与安慰剂相比,44名肥胖或超重女性接受80mg盐酸吡哆醇(维生素B6的主要形式)治疗8周,胰岛素敏感性提高,脂肪量减少25。PLP的补充显著降低了糖尿病大鼠中高浓度的糖基化诱导的毒性化合物,并阻止了糖尿病肾病的进展21,26。
最近的一项研究进一步确定,接受PLP治疗的糖尿病大鼠与糖尿病相关的问题有所减少,包括氧化应激参数下降、血糖水平下降以及肝脏和肾脏损伤恢复27。此外,吡哆胺(Pyridoxamine维生素B6的另一种形式)改善了肥胖2型糖尿病小鼠的胰岛素敏感性28。已知吡哆胺可抑制AGEs和活性氧形成等29,30。
2.抑制神经病变:
2.1.硫辛酸:
它兼具水溶性和脂溶性,可在体内产生并发挥多种生物学作用,尤其是在葡萄糖代谢方面31。事实上,硫辛酸在德国已被批准用于治疗糖尿病神经病变31,32。此外,硫辛酸可能对糖尿病患者的葡萄糖代谢产生积极影响33。硫辛酸帮助对抗糖尿病神经病变的重要机制包括抑制糖化和炎症34,45。据报道,硫辛酸可以在神经元细胞培养中防止氧化损伤36。它还改善了神经的血液流动,使它们能够更有效地利用能量31,37。
一项临床试验表明,在3周内每天3次服用600mg的α-硫辛酸,可显著改善12名糖尿病神经病变患者的症状。在另一项试验中,181名患者被分为每天接受安慰剂或600、1200或1800mg α-硫辛酸组。1800mg/天组在短短一周内症状显著改善,600mg和1200mg组在第2周时症状有所改善31。在这两项试验中,疼痛和灼烧感都得到了缓解。另一项研究发现,在4年的时间里,每天口服600mg硫辛酸可以改善神经性症状,减缓轻度至中度糖尿病神经病变的进展,阻止其进一步恶化38。总之,口服硫辛酸可缓解疼痛、灼烧麻木感等,减少肌无力,提高整体生活质量39,40。
其他试验表明,静脉注射硫辛酸对糖尿病神经病变也有益。一项研究表明,每天静脉注射α-硫辛酸3周是治疗糖尿病神经病变的有效方法,另一项试验报告称,静脉注射2-4周是有效的31,41。
注意:硫辛酸有二种形式的产品:α-硫辛酸(含s-和r-型硫辛酸各占50%)和r-硫辛酸。有证据表明,r-硫辛酸的钠盐比a-硫辛酸更具生物利用性42。
2.2.乙酰L肉碱(ALC)和L肉碱:
肉碱是一种对线粒体能量产生很重要的氨基酸类化合物。对肉碱在糖尿病神经病变中的作用源于有证据表明,患有并发症(包括糖尿病神经病变)的糖尿病患者的血清和总肉碱水平低于没有并发症的糖尿病患者43。因此,ALC和L-肉碱均已被研究为糖尿病神经病变的潜在治疗方法。补充肉碱可能有助于对抗肉碱缺乏症,改善胰岛素抵抗,使细胞更有效地利用葡萄糖,帮助受损神经纤维再生,或帮助受损神经元更有效地运输细胞内成分43。
已经对补充肉碱对糖尿病神经病变的影响进行了多项人体试验。一项研究发现,L-肉碱(每天2g,持续10个月)可以改善神经传导速度,而神经传导速度在糖尿病神经病变中受损44。对ALC的研究发现,它减轻了糖尿病神经病变患者的疼痛,改善了腿部的振动感,并增加了神经再生45-47。补充肉碱也可能有助于治疗糖尿病引起的自主神经病变,对动物模型进行的一项研究发现ALC降低了糖尿病自主神经病变的心血管体征48。
ALC对外周神经系统具有神经保护和镇痛作用49;可防止神经细胞死亡、促进神经再生和神经损伤修复以及增强对神经生长因子的反应等50。在糖尿病患者中,剂量为1500mg/天至3000mg/天的乙酰-L-肉碱可提高神经传导速度和强度,降低疼痛和残疾评分,增加神经纤维数量,并再生受损的神经纤维51,52,46。
2.3.十六酰胺乙醇(PEA):
一种天然存在于全身组织中的脂质化合物,包括中枢神经系统。在大豆卵磷脂、蛋黄和花生等食物中也存在53。越来越多的研究表明,补充PEA可能有效缓解各种原因引起的疼痛,而不会引发不良副作用54,55。大多数现有的临床前研究表明,PEA通过改变某些基因的表达和减少炎症信号传导发挥作用,但也提出了其镇痛作用的其他可能机制,包括其通过神经系统中的大麻素受体刺激信号传导的能力56-58。
几项临床试验表明,PEA可以减轻多种原因引起的疼痛,包括糖尿病神经病变、化疗诱导的周围神经病变、坐骨神经压迫、腕管综合征、骨关节炎、腰痛、背部手术失败、中风相关神经痛、多发性硬化症、牙痛、慢性骨盆疼痛、疱疹后神经痛和阴道疼痛等59。在一项针对因各种情况而无法通过常规治疗控制疼痛的慢性疼痛患者的观察性研究中,在三周内每天二次添加600mg PEA,然后在四周内每天一次,降低了所有完成研究的参与者的平均疼痛强度得分60。
在一项随机对照试验中,636名因坐骨神经压迫而疼痛的参与者接受了每天300mg PEA、每天600mg PEA或安慰剂以及他们常用的止痛药,为期三周。两种剂量的PEA都比安慰剂更能减轻疼痛,而且高剂量比低剂量更有效。事实上,600mg组的疼痛评分降低了50%以上61。在118名神经疼痛患者中,30天的标准治疗加上每天600mg PEA比单独的标准治疗更有效62。一项随机临床试验发现,在缓解颞下颌关节(TMJ)疼痛方面,一周内每天900mg PEA,一周后每天600mg PEA比布洛芬更有效,布洛芬的剂量为600mg,两周内每天三次63。
对PEA的微粉化制剂也进行了研究。微粉化会产生更小的颗粒,这些颗粒可能更容易被吸收。剂量为600–1200mg/天的微粉化PEA可减轻糖尿病或创伤相关神经疼痛、背部手术失败后的慢性疼痛和拔牙后的急性疼痛受试者的疼痛64-66。在一份关于100例与脊柱疾病相关的神经疼痛的报告中,将超微粉化PEA补充剂纳入疼痛管理治疗显示出了有希望的结果67。一项荟萃分析发现,患有子宫内膜异位症引起的慢性盆腔疼痛的女性似乎受益于每天800mg微粉化PEA加上每天80mg虎杖苷(Polydatin)的组合68。在一项随机对照试验中,PEA和虎杖苷的组合在减轻肠易激综合征患者腹痛方面比安慰剂更有效69。
2.4. N-乙酰半胱氨酸(NAC):
既是体内谷胱甘肽合成前体,NAC本身也是一种抗氧化剂。它可以保护神经元免受氧化损伤70。此外,NAC已被证明可以抑制AGEs的形成71。对糖尿病神经病变动物模型的多项研究发现,NAC可以防止神经元死亡并防止神经损伤70,32。
2.5.维生素B族:
B族维生素是一种维生素家族,在人体中发挥着许多作用,尤其是在细胞能量生成和神经系统功能中72。
2.6.姜黄素:
姜黄素因其抗炎特性而著称,最近也因其在疼痛控制方面的潜力而受到研究88,89。事实上,姜黄素已被证明可以显著提高实验动物的疼痛阈值并降低疼痛超敏反应90-92。
对于患有糖尿病神经损伤的人来说,姜黄素提供的不仅仅是止痛,也可能减缓或逆转最初产生神经性疼痛的一些过程。临床前研究表明,姜黄素可减少TNF-α的产生,TNF-α是一种炎症蛋白,也是疼痛的诱因91,激活内部疼痛缓解系统(称为内源性阿片系统)89;减少细胞中的氧化应激,这是神经疼痛的主要诱因92,以及减少糖尿病神经中的异常电脉冲93,94。
其他实验研究和人体试验表明,姜黄素是一种很有前途的预防和治疗糖尿病及其并发症的天然药物,主要作用机制是中和反应性自由基和减少炎症的能力。姜黄素似乎可以提高胰岛素敏感性,降低血液中的葡萄糖和脂质水平。它还可以保护胰腺中产生胰岛素的β细胞95-97。
3.降低血管损伤、改善内皮功能:
3.1.碧萝芷:
在碧萝芷与代谢综合征(包括高血压、糖尿病等)的综述中发现,它有助于改善血糖指标、降低血压值等98。碧萝芷已被研究用于治疗糖尿病视网膜病变99,它可以保护视网膜中的毛细血管免受损伤。
一项研究77名2型糖尿病患者的临床试验检测了碧萝芷对血管损伤标志物内皮素-1水平的影响,内皮素-1是血管损伤的标志。34名受试者在12周内每天服用100mg碧萝芷,与43名服用安慰剂的受试者相比,内皮素-1水平降低100。一项对1289人进行的多项研究的全面综述发现,碧萝芷对治疗糖尿病视网膜病变可能有用。这篇文章的结论是,60至150mg的碧萝芷剂量有助于减缓糖尿病视网膜病变的进展,提高2型糖尿病患者的视力,提高毛细血管的强度,并减少毛细血管渗漏到视网膜中101。
另一项研究考察了在二个月内每天给2型糖尿病患者服用150mg碧萝芷的效果,发现24名接受碧萝芷治疗的受试者与接受安慰剂治疗的22名受试者相比,视力提高,视网膜肿胀和增厚减少102。除了含有可以中和糖尿病引起的视网膜损伤的化合物外,碧萝芷中的一些化合物可能有助于修复受损的毛细血管99。
2型糖尿病患者合并有相当大的心血管疾病(CVD)的过度发病和死亡风险。一项金标准临床试验(RCT)包括48名受试者,旨在测试碧萝芷在减少2型糖尿病患者降压药物使用和心血管疾病危险因素方面的临床有效性。补充碧萝芷12周后,与对照组相比,碧萝芷改善了糖尿病控制,降低了心血管疾病风险因素,并减少了抗高血压药物的使用103。
3.2.维生素E(γ-生育酚):
餐后血糖飙升会损伤血管内壁,导致内皮功能障碍和血管疾病。γ-生育酚是维生素E的一种形式,具有抗炎和清除自由基的活性。在健康男性中进行的两项试验发现,补充γ-生育酚可以防止餐后葡萄糖峰值诱导的内皮功能障碍相关变化104,105。
3.3葡萄籽提取物:
所含原花青素可调节氧化应激,并已被研究用于多种健康状况,包括高血压、癌症和阿尔茨海默病106-108。葡萄多酚似乎具有重要的抗糖尿病作用,并保护组织免受血糖升高的损害。
在一项针对32名2型糖尿病患者的为期四周的随机安慰剂对照试验中,与安慰剂相比,每天摄入600mg葡萄籽提取物可显著降低果糖胺(Fructosamine)。果糖胺是一种类似于HbA1C的测试,可以测量几周内的血糖水平。葡萄籽提取物还降低了总胆固醇和Hs-CRP,并显著提高了血液谷胱甘肽-人体主要的内源性抗氧化剂之一109。另一项试验发现,红酒中富含葡萄多酚的非酒精成分可以提高胰岛素敏感性,降低心血管风险110。
一项随机对照试验,每天给2型糖尿病的健康但超重的一级亲属服用2g葡萄多酚,为期8周。然后,受试者被大量摄入果糖(果汁和许多加糖饮料中的糖)。在安慰剂组中,果糖激发导致氧化应激增加,胰岛素敏感性和线粒体活性降低。多酚组预防了所有这些负面影响111。
4.维持基础营养:
4.1.辅酶Q10(CoQ10):
CoQ10对线粒体能量代谢至关重要,也是氧化应激的强大抑制剂112。辅酶Q10缺乏与糖尿病有关113,114。在一项针对64名2型糖尿病患者的随机对照试验中,连续12周每天补充200mg辅酶Q10可降低血清HbA1C浓度,并降低总胆固醇和LDL胆固醇水平114。一项针对74名2型糖尿病受试者的临床试验发现,每天两次100mg辅酶Q10可显著降低HbA1C和血压值115。在一项针对23名他汀类药物治疗的2型糖尿病患者的安慰剂对照试验中,每天200mg辅酶Q10显著改善了糖尿病患者血管内皮功能障碍的标志物116。
在糖尿病动物模型中,辅酶Q10治疗显著改善了胰岛素抵抗,降低了血清胰岛素和葡萄糖水平,并将能量调节激素脂联素水平提高了六倍113。高水平的脂联素与糖尿病和心血管并发症的风险降低有关117,118。
动物研究表明,长期使用辅酶Q10对进行性糖尿病神经病变具有保护作用。辅酶Q10的有益作用可能归因于减少氧化损伤和炎症,这两个因素都与糖尿病神经病变有关119。
还原形式的辅酶Q10泛醇比泛醌形式更有效地被吸收,对中老年人尤其适应120,121。
4.2.维生素D:
许多研究证实,维生素D缺乏(低于20ng/mL)和不足(20ng/mL至30ng/mL)与与糖尿病患者的神经病变密切相关122-125。维生素D状态已被证明与葡萄糖代谢有关126,127。在不同人群中,较高水平的血清维生素D被证明可以降低糖尿病风险128。
慢性全身炎症是胰岛素抵抗和糖尿病的一个特征,可能是一种影响,但有人认为它实际上可能是病因127,129。负责炎症的免疫细胞表达维生素D受体,维生素D在多种情况下成功抑制慢性炎症,而维生素D缺乏与促炎状态有关129,130。
一项研究发现,维生素D水平不足的糖尿病患者患神经性疾病的几率高出2倍124。临床试验已经证实,每周50,000IU的维生素D3可以显著减轻糖尿病神经损伤的症状,提高生活质量131,132。在一项研究中,每二周服用50,000IU的维生素D,12周后糖尿病足溃疡的面积显著缩小133。
一般认为,维持血清25-羟基维生素D水平的最佳目标范围在50至80ng/mL之间。
4.3.欧米伽3脂肪酸:
鱼油和亚麻籽油富含抗炎的ω-3脂肪酸。增加ω-3脂肪酸摄入在预防和缓解糖尿病神经病变方面显示出巨大的前景。在啮齿类动物研究中,鱼油可以减缓甚至逆转糖尿病神经损伤的进展134,135。
研究表明,ω-3脂肪酸可能有助于治疗糖尿病神经病变路径包括:糖尿病神经病变患者的神经传导速度下降,神经传导速度衡量电脉冲在神经中的移动速度。动物研究发现,鱼油能迅速恢复神经传导速度,减少对关键神经束的可见损伤136;使用鱼油治疗后,糖尿病动物的疼痛和对触摸的超敏反应减轻,动物的神经簇显示炎症减少,核因子κB(NF-kB,主要促炎因子)水平降低137;在用DHA或EPA处理后,实验室培养的神经细胞会急剧增加其对抗有害氧化应激的蛋白质产量138。
上述不同的作用会产生重大后果。在一项针对糖尿病足溃疡(可导致截肢的神经病变并发症)患者的研究中,每天服用1g ω-3可显著缩小溃疡面积。与此同时,炎症标志物降低,总抗氧化水平显著升高139。
此外,鱼油中的DHA和EPA似乎可以防止与餐后血糖飙升相关的一些血管功能变化。一项针对34名2型糖尿病受试者的为期六周的试验发现,补充ω-3脂肪酸可以显著防止餐后小血管和大血管功能障碍140。
4.4.烟酰胺腺嘌呤二核苷酸(NAD+):
烟酰胺腺嘌呤二核苷酸(NAD+)是细胞能量的关键调节因子141。它也是Sirtuin蛋白的辅因子,该蛋白参与许多代谢活动,并与寿命有关。衰老与SIRT1活性下降有关,SIRT1是编码Sirtuin 1蛋白的基因。临床前研究表明,SIRT1表达增加可延长寿命142。与年龄相关的NAD+水平下降与SIRT1活性降低有关143,144。NAD+代谢也与糖尿病的病因和并发症有关145-147。
众所周知,烟酰胺核糖苷和NMN(β-烟酰胺单核苷酸)都是维生素B3衍生物,补充可以提高细胞NAD+水平。
4.5.牛磺酸:
牛磺酸是一种遍布全身的氨基磺酸,且在大脑、心脏和骨骼肌中含量特别丰富。牛磺酸参与多种重要的生理功能,如促进细胞膜稳定和促进健康的神经系统功能148。临床前研究发现,牛磺酸可能会抵消一些导致糖尿病并发症的问题(如糖尿病肾病、糖尿病白内障和糖尿病心肌病)149。观察性研究发现,与非糖尿病对照组相比,糖尿病患者的血浆牛磺酸浓度往往较低150,151。
一项荟萃分析评估了五项随机对照试验中补充牛磺酸对血糖标志物的影响,该试验共招募了209名参与者。在这些研究中,糖尿病患者每天接受500-3000mg牛磺酸或安慰剂治疗,持续2-16周。与对照组相比,那些服用牛磺酸的人降低了HbA1C和空腹血糖,并在胰岛素抵抗模型中有所改善152。
在荟萃分析中的一项研究中,当牛磺酸与全身阻力运动暂停训练的独特力量训练相结合时,患有2型糖尿病的女性的改善最大。这种训练形式允许使用体重和重力作为阻力进行单关节和多关节训练。当每天两次(早、晚)服用500mg牛磺酸,持续八周,并与此训练相结合时,与只服用一种或另一种或安慰剂的组相比,参与者的体脂百分比和胰岛素抵抗降低幅度更大,高密度脂蛋白增加幅度更大153。这些发现表明,补充牛磺酸与力量训练相结合可能是支持糖尿病患者代谢健康的一种很有前途的干预措施。
以及可参阅本网站如下专文的相关内容:
参考文献:
1. Ahmed N et al. Advanced glycation endproducts: what is their relevance to diabetic complications? Diabetes Obes Metab. 2007 May;9(3):233-45.
2. Sugimoto K et al. Role of advanced glycation end products in diabetic neuropathy. Curr Pharm Des. 2008;14(10):953-61.
3. Fournet M et al. Glycation Damage: A Possible Hub for Major Pathophysiological Disorders and Aging. Aging Dis. 2018 Oct;9(5):880-900.
4. Vincent AM et al. Diabetic neuropathy: cellular mechanisms as therapeutic targets. Nat Rev Neurol. 2011 Sep 13;7(10):573-83.
5. Beltramo E et al. Effects of thiamine and benfotiamine on intracellular glucose metabolism and relevance in the prevention of diabetic complications. Acta Diabetol. 2008 Sep;45(3):131-41.
6. Balakumar P et al. The multifaceted therapeutic potential of benfotiamine. Pharmacol Res. 2010 Jun;61(6):482-8.
7. Xie F et al. Pharmacokinetic study of benfotiamine and the bioavailability assessment compared to thiamine hydrochloride. J Clin Pharmacol. 2014 Jun;54(6):688-95.
8. Hammes H-A et al. Benfotiamine blocks three major pathways of hyperglycemic damage and prevents experimental diabetic retinopathy. Nat Med. 2003 Mar;9(3):294-9.
9. Shoeb M et al. Anti-inflammatory effects of benfotiamine are mediated through the regulation of the arachidonic acid pathway in macrophages. Free Radic Biol Med. 2012 Jan 1;52(1):182-90.
10. Stracke H et al. Benfotiamine in diabetic polyneuropathy (BENDIP): results of a randomized, double blind, placebo-controlled clinical study. Exp Clin Endocrinol Diabetes. 2008 Nov;116(10):600-5.
11. Winkler G, Pal B, Nagybeganyi E, et al. Effectiveness of different benfotiamine dosage regimens in the treatment of painful diabetic neuropathy. Arzneimittelforschung. 1999 Mar;49(3):220-4.
12. Stirban A et al. Benfotiamine prevents macro- and microvascular endothelial dysfunction and oxidative stress following a meal rich in advanced glycation end products in individuals with type 2 diabetes. Diabetes Care. 2006 Sep;29(9):2064-71.
13. Nikolic A et al. [The effect of benfothiamine in the therapy of diabetic polyneuropathy]. Srp Arh Celok Lek. 2009 Nov-Dec;137(11-12):594-600.
14. Chakrabarti R et al. Preventive effects of benfotiamine in chronic diabetic complications. J Diabetes Investig. 2011 Apr 7;2(2):123-31.
15. Haupt E et al. Benfotiamine in the treatment of diabetic polyneuropathy--a three-week randomized, controlled pilot study (BEDIP study). Int J Clin Pharmacol Ther. 2005 Feb;43(2):71-7.
16. Boldyrev AA et al. Carnosine as a natural antioxidant and geroprotector: from molecular mechanisms to clinical trials. Rejuvenation Res. 2010 Apr-Jun;13(2-3):156-8.
17. Brown BE et al. Supplementation with carnosine decreases plasma triglycerides and modulates atherosclerotic plaque composition in diabetic apo E(-/-) mice. Atherosclerosis. 2014 Feb;232(2):403-9.
18. Ansurudeen I et al. Carnosine enhances diabetic wound healing in the db/db mouse model of type 2 diabetes. Amino Acids. 2012 Jul;43(1):127-34.
19. Yapislar H et al. Effect of carnosine on erythrocyte deformability in diabetic rats. Arch Physiol Biochem. 2012 Dec;118(5):265-72.
20. Mascolo E et al. Vitamin B6 and Diabetes: Relationship and Molecular Mechanisms. Int J Mol Sci. 2020 May 23;21(10):3669.
21. Higuchi O et al. Aminophospholipid glycation and its inhibitor screening system: a new role of pyridoxal 5'-phosphate as the inhibitor. J Lipid Res. 2006 May;47(5):964-74.
22. Fishman SL et al. The role of advanced glycation end-products in the development of coronary artery disease in patients with and without diabetes mellitus: a review. Mol Med. 2018 Nov 23;24(1):59.
23. Horikawa C et al. Vitamin B6 intake and incidence of diabetic retinopathy in Japanese patients with type 2 diabetes: analysis of data from the Japan Diabetes Complications Study (JDCS). Eur J Nutr. 2020 Jun;59(4):1585-1594.
24. Walker KZ et al. Diet and exercise in the prevention of diabetes. J Hum Nutr Diet. 2010 Aug;23(4):344-52.
25. Haidari E et al. The Effect of Pyridoxine Hydrochloride Supplementation on Leptin, Adiponectin, Glycemic Indices, and Anthropometric Indices in Obese and Overweight Women. Clin Nutr Res. 2021 Jul 22;10(3):230-242.
26. Nakamura S et al. Pyridoxal phosphate prevents progression of diabetic nephropathy. Nephrol Dial Transplant. 2007 Aug;22(8):2165-74.
27. Abdullah KM et al. Anti-diabetic study of vitamin B6 on hyperglycaemia induced protein carbonylation, DNA damage and ROS production in alloxan induced diabetic rats. Toxicol Res (Camb). 2019 Jun 11;8(4):568-579.
28. Mastrocola R et al. Altered hepatic sphingolipid metabolism in insulin resistant mice: Role of advanced glycation endproducts. Free Radic Biol Med. 2021 Jun:169:425-435.
29. Abdullah KM et al. Study of pyridoxamine against glycation and reactive oxygen species production in human serum albumin as model protein: An in vitro & ex vivo approach. Int J Biol Macromol. 2018 Dec;120(Pt B):1734-1743.
30. Ramis R et al. How Does Pyridoxamine Inhibit the Formation of Advanced Glycation End Products? The Role of Its Primary Antioxidant Activity. Antioxidants (Basel). 2019 Sep 1;8(9):344.
31. McIlduff CE et al. Critical Appraisal of the Use of Alpha Lipoic Acid (Thiooctic Acid) in the Treatment of Symptomatic Diabetic Polyneuropathy. Therapeutics and Clinical Risk Management, 2011; 7: 377-385.
32. Head KA. Peripheral Neuropathy: Pathogenic Mechanisms and Alternative Therapies. Alternative Medicine Review, 2006; 11(4): 294-329.
33. Korotchkina LG et al. R-lipoic acid inhibits mammalian pyruvate dehydrogenase kinase. Free radical research. Oct 2004;38(10):1083-1092.
34. Thirunavukkarasu V et al. Lipoic acid improves glucose utilisation and prevents protein glycation and AGE formation. Die Pharmazie. Oct 2005;60(10):772-775.
35. Kunt T et al. Alpha-lipoic acid reduces expression of vascular cell adhesion molecule-1 and endothelial adhesion of human monocytes after stimulation with advanced glycation end products. Clinical science. Jan 1999;96(1):75-82.
36. Bharat S et al. Pre-treatment with R-lipoic acid alleviates the effects of GSH depletion in PC12 cells: implications for Parkinson's disease therapy. Neurotoxicology. Oct 2002;23(4-5):479-486.
37. Bertolotto F et al. Combination of Alpha Lipoic Acid and Superoxide Dismutase Leads to Physiological and Symptomatic Improvements in Diabetic Neuropathy. Drugs Research and Development, 2012; 12(1): 29-34.
38. Ziegler D et al. Efficacy and Safety of Antioxidant Treatment with Alpha-Lipoic Acid Over 4 Years in Diabetic Polyneuropathy. Diabetes Care, 2011; 34: 2054-2060.
39. Garcia-Alcala H et al. Treatment with alpha-Lipoic Acid over 16 Weeks in Type 2 Diabetic Patients with Symptomatic Polyneuropathy Who Responded to Initial 4-Week High-Dose Loading. J Diabetes Res. 2015;2015:189857.
40. Agathos E et al. Effect of alpha-lipoic acid on symptoms and quality of life in patients with painful diabetic neuropathy. J Int Med Res. 2018 May;46(5):1779-90.
41. Han T et al. A Systematic Review and Meta-Analysis of Alpha-Lipoic Acid in the Treatment of Diabetic Peripheral Neuropathy. European Journal of Endocrinology, 2012; 167: 465-471.
42. Carlson DA et al. The plasma pharmacokinetics of R-(+)-lipoic acid administered as sodium R-(+)-lipoate to healthy human subjects. Alternative medicine review. Dec 2007;12(4):343-351.
43. Evans JD et al. Role of Acetyl-L-Carnitine in the Treatment of Diabetic Peripheral Neuropathy. Annals of Pharmacotherapy, 2008; 42: 1686-1691.
44. Ulvi H et al. Effect of L-Carnitine on Diabetic Neuropathy and Ventricular Dispersion in Patients with Diabetes Mellitus. Turkish Journal of Medical Science, 2010; 40(2): 169-175.
45. Bansal V et al. Diabetic Neuropathy. Postgraduate Medical Journal, 2006; 82: 95-100.
46. Sima AA et al. Acetyl-L-Carnitine Improves Pain, Nerve Regeneration, and Vibratory Perception in Patients with Chronic Diabetic Neuropathy. Diabetes Care, 2005; 28(1): 89-94.
47. Adriaensen H et al. Critical Review of Oral Drug Treatments for Diabetic Neuropathic Pain – Clinical Outcomes Based on Efficacy and Safety Data from Placebo-Controlled and Direct Comparative Studies. Diabetes/Metabolism Research and Reviews, 2005; 21: 231-240.
48. Giudice PL et al. Autonomic Neuropathy in Streptozotocin Diabetic Rats: Effect of Acetyl-L-Carnitine. Diabetes Research and Clinical Practice, 2002; 56: 173-180.
49. Onofrj M et al. Acetyl-L-carnitine: from a biological curiosity to a drug for the peripheral nervous system and beyond. Expert Rev Neurother. 2013 Aug;13(8):925-36.
50. Sergi G et al. Effects of acetyl-L-carnitine in diabetic neuropathy and other geriatric disorders. Aging Clin Exp Res. 2018 Feb;30(2):133-8.
51. De Grandis D et al. Acetyl-L-carnitine (levacecarnine) in the treatment of diabetic neuropathy. A long-term, randomised, double-blind, placebo-controlled study. Drugs R D. 2002;3(4):223-31.
52. Li S et al. Effects of acetyl-L-carnitine and methylcobalamin for diabetic peripheral neuropathy: A multicenter, randomized, double-blind, controlled trial. J Diabetes Investig. 2016 Sep;7(5):777-85.
53. Mattace Raso G et al. Palmitoylethanolamide in CNS health and disease. Pharmacological Research. Aug 2014;86:32-41.
54. Artukoglu BB et al. Efficacy of Palmitoylethanolamide for Pain: A Meta-Analysis. Pain Physician. Jul 2017;20(5):353-362.
55. Paladini A et al. Palmitoylethanolamide, a Special Food for Medical Purposes, in the Treatment of Chronic Pain: A Pooled Data Meta-analysis. Pain Physician. Feb 2016;19(2):11-24.
56. Gabrielsson L et al. Palmitoylethanolamide for the treatment of pain: pharmacokinetics, safety and efficacy. British Journal of Clinical Pharmacology. Oct 2016;82(4):932-942.
57. Khasabova IA et al. Peroxisome proliferator-activated receptor alpha mediates acute effects of palmitoylethanolamide on sensory neurons. The Journal of Neuroscience. Sep 12 2012;32(37):12735-12743.
58. Di Cesare Mannelli L et al. Palmitoylethanolamide is a disease-modifying agent in peripheral neuropathy: pain relief and neuroprotection share a PPAR-alpha-mediated mechanism. Mediators of Inflammation. 2013;2013:328797.
59. Keppel Hesselink JM et al. Therapeutic utility of palmitoylethanolamide in the treatment of neuropathic pain associated with various pathological conditions: a case series. Journal of Pain Research. 2012;5:437-442.
60. Gatti A et al. Palmitoylethanolamide in the treatment of chronic pain caused by different etiopathogenesis. Pain Medicine (Malden, Mass.). Sep 2012;13(9):1121-1130.
61. Keppel Hesselink JM et al. Palmitoylethanolamide, a neutraceutical, in nerve compression syndromes: efficacy and safety in sciatic pain and carpal tunnel syndrome. Journal of Pain Research. 2015;8:729-734.
62. Dominguez CM et al. N-palmitoylethanolamide in the treatment of neuropathic pain associated with lumbosciatica. Pain Management. Mar 2012;2(2):119-124.
63. Marini I et al. Palmitoylethanolamide versus a nonsteroidal anti-inflammatory drug in the treatment of temporomandibular joint inflammatory pain. Journal of Orofacial Pain. Spring 2012;26(2):99-104.
64. Cocito D et al. Short-term efficacy of ultramicronized palmitoylethanolamide in peripheral neuropathic pain. Pain Research and Treatment. 2014;2014:854560.
65. Paladini A et al. Palmitoylethanolamide in the Treatment of Failed Back Surgery Syndrome. Pain Research and Treatment. 2017;2017:1486010.
66. Bacci C et al. Randomized split-mouth study on postoperative effects of palmitoylethanolamide for impacted lower third molar surgery. ISRN Surgery. 2011;2011:917350.
67. Chirchiglia D et al. Nonsurgical lumbar radiculopathies treated with ultramicronized palmitoylethanolamide (umPEA): A series of 100 cases. Neurol Neurochir Pol. 2018 Jan-Feb;52(1):44-47.
68. Indraccolo U et al. Micronized palmitoylethanolamide/trans-polydatin treatment of endometriosis-related pain: a meta-analysis. Annali Dell'Istituto Superiore di Sanita. Apr-Jun 2017;53(2):125-134.
69. Cremon C et al. Randomised clinical trial: the analgesic properties of dietary supplementation with palmitoylethanolamide and polydatin in irritable bowel syndrome. Alimentary Pharmacology & Therapeutics. Apr 2017;45(7):909-922.
70. Kamboj SS et al. N-Acetylcysteine Inhibits Hyperglycemia-Induced Oxidative Stress and Apoptosis Markers in Diabetic Neuropathy. Journal of Neurochemistry, 2010; 112: 77-91.
71. Nakayama M et al. Suppression of N(epsilon)-(carboxymethyl)lysine generation by the antioxidant N-acetylcysteine. Peritoneal dialysis international. May-Jun 1999;19(3):207-210.
72. Selhub J et al. B vitamins, homocysteine, and neurocognitive function in the elderly. The American journal of clinical nutrition. Feb 2000;71(2):614s-620s.
73. Sanchez-Ramirez GM et al. Benfotiamine Relieves Inflammatory and Neuropathic Pain in rats. European Journal of Pharmacology, 2006; 530: 48-53.
74. Varkonyi T et al. Diabetic Neuropathy: New Strategies for Treatment. Diabetes, Obesity and Metabolism, 2008; 10: 99-108.
75. Fahmy E et al. Estimation of Serum Homocysteine Level in Patients with Type 2 Diabetic Neuropathy. Egyptian Journal of Neurology Psychiatry and Neurosurgery, 2010; 47(1): 59-66.
76. Mizukami H et al. Methylcobalamin Effects of Diabetic Neuropathy and Nerve Protein Kinase C in Rats. European Journal of Clinical Investigation, 2011; 41(4): 442-450.
77. Okada K et al. Methylcobalamin Increases Erk1/2 and Akt Activities through the Methylation Cycle and Promotes Nerve Regeneration in a Rat Sciatic Nerve Injury Model. Experimental Neurology, 2010;222: 191-203.
78. Jacobs AM et al. Management of Diabetic Small-Fiber neuropathy with Combination L-Methylfolate, Methylcobalamin, and Pyridocal 5’-Phosphate. Reviews of Neurological Diseases, 2011; 8(1/2): 1-9.
79. Fonseca VA et al. Metanx in Type 2 Diabetes with Peripheral Neuropathy: A Randomized Trial. The American Journal of Medicine (2013) 126, 141-149.
80. Wade RL et al. Impact of L-Methylfolate Combination therapy Among Diabetic Peripheral Neuropathy Patients. American Journal of Pharmacological Benefits, 2012; 4(5): 218-225.
81. Talaei A et al. Vitamin B12 may be more effective than nortriptyline in improving painful diabetic neuropathy. International Journal of Food Sciences and Nutrition, 2009;60 Suppl 5:71-6.
82. Dominguez JC et al. A prospective, open label, 24-week trial of methylcobalamin in the treatment of diabetic polyneuropathy. Journal of Diabetes Mellitus, 2012; 2(4): 408-412.
83. Scaglione F et al. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica. 2014 May;44(5):480-8.
84. Bhatia P et al. Homocysteine excess: delineating the possible mechanism of neurotoxicity and depression. Fundam Clin Pharmacol. 2015 Dec;29(6):522-8.
85. Ambrosch A et al. Relation between homocysteinaemia and diabetic neuropathy in patients with Type 2 diabetes mellitus. Diabet Med. 2001 Mar;18(3):185-92.
86. Wang D et al. Serum folate, vitamin B12 levels and diabetic peripheral neuropathy in type 2 diabetes: A meta-analysis. Mol Cell Endocrinol. 2017 Mar 5;443:72-9.
87. Mottaghi T et al. Effect of folic acid supplementation on nerve conduction velocity in diabetic polyneuropathy patients. Neurol Res. 2019 Apr;41(4):364-8.
88. Sun J et al. Role of curcumin in the management of pathological pain. Phytomedicine. 2018 Sep 15;48:129-40.
89. Banafshe HR et al. Effect of curcumin on diabetic peripheral neuropathic pain: possible involvement of opioid system. Eur J Pharmacol. 2014 Jan 15;723:202-6.
90. Attia HN et al. Protective effects of combined therapy of gliclazide with curcumin in experimental diabetic neuropathy in rats. Behav Pharmacol. 2012 Apr;23(2): 153-61.
91. Li Y et al. Curcumin attenuates diabetic neuropathic pain by downregulating TNF-alpha in a rat model. Int J Med Sci. 2013;10(4):377-81.
92. Zhao WC et al. Curcumin ameliorated diabetic neuropathy partially by inhibition of NADPH oxidase mediating oxidative stress in the spinal cord. Neurosci Lett. 2014 Feb 7;560:81-5.
93. Meng B et al. Effects of curcumin on TTX-R sodium currents of dorsal root ganglion neurons in type 2 diabetic rats with diabetic neuropathic pain. Neurosci Lett. 2015 Sep 25;605: 59-64.
94. Meng B et al. Effects of curcumin on sodium currents of dorsal root ganglion neurons in type 2 diabetic neuropathic pain rats. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2015 Nov;31(6):541-8.
95. Nabavi SF et al. Curcumin: a natural product for diabetes and its complications. Curr Top Med Chem. 2015;15(23):2445-55.
96. Zhang DW et al. Curcumin and diabetes: a systematic review. Evid Based Complement Alternat Med. 2013;2013:636053
97. Meng B et al. Antioxidant and antiinflammatory activities of curcumin on diabetes mellitus and its complications. Curr Pharm Des. 2013;19(11):2101-13.
98. Gulati OP. Pycnogenol® in Metabolic Syndrome and Related Disorders. Phytother Res. 2015 Jul;29(7):949-68.
99. Spadea L et al. Treatment of Cascular Retinopathies with Pycnogenol. Phytotherapy Research, 2001; 15: 219-223.
100. Liu X et al. Antidiabetic effect of PycnogenolR French maritime pine bark extract in patients with diabetes type II. Life Sciences, 2004; 75: 2505-2513.
101. Schonlau F et al. Pycnogenol for Diabetic Retinopathy. International Ophthalmology, 2002; 24: 161-171.
102. Steigerwalt R et al. Pycnogenol® Improves Microcirculation, Retinal Edema, and Visual Acuity in Early Diabetic Retinopathy. Journal of Ocular Pharmacology and Therapeutics, 2009; 25(6): 537-540.
103. Zibadi S et al. Reduction of cardiovascular risk factors in subjects with type 2 diabetes by Pycnogenol supplementation. Nutr Res. 2008 May;28(5):315-20.
104. Mah E et al. Supplementation of a γ-tocopherol-rich mixture of tocopherols in healthy men protects against vascular endothelial dysfunction induced by postprandial hyperglycemia. J Nutr Biochem. 2013 Jan;24(1):196-203.
105. Masterjohn C et al. γ-Tocopherol abolishes postprandial increases in plasma methylglyoxal following an oral dose of glucose in healthy, college-aged men. J Nutr Biochem. 2012 Mar;23(3):292-8.
106. Pasinetti GM et al. Role of grape seed polyphenols in Alzheimer’s disease neuropathology. Nutr Diet Suppl. 2010 Aug 1; 2010(2): 97–103.
107. Feringa HHH et al. The effect of grape seed extract on cardiovascular risk markers: a meta-analysis of randomized controlled trials. J Am Diet Assoc. 2011 Aug;111(8):1173-81.
108. Kaur M et al. Anticancer and Cancer Chemopreventive Potential of Grape Seed Extract and Other Grape-Based Products. J Nutr. 2009 Sep; 139(9): 1806S–1812S.
109. Kar P et al. Effects of grape seed extract in Type 2 diabetic subjects at high cardiovascular risk: a double blind randomized placebo controlled trial examining metabolic markers, vascular tone, inflammation, oxidative stress and insulin sensitivity. Diabet Med. 2009 May;26(5):526-31.
110. Chiva Blanch G et al. Effects of red wine polyphenols and alcohol on glucose metabolism and the lipid profile: a randomized clinical trial. Clin Nutr. 2013 Apr;32(2):200-6.
111. Hokayem M et al. Grape polyphenols prevent fructose-induced oxidative stress and insulin resistance in first-degree relatives of type 2 diabetic patients. Diabetes Care. 2013 Jun;36(6):1454-61.
112. Littarru GP et al. Bioenergetic and antioxidant properties of coenzyme Q10: recent developments. Mol Biotechnol. 2007 Sep;37(1):31-7.
113. Amin MM et al. Novel CoQ10 antidiabetic mechanisms underlie its positive effect: modulation of insulin and adiponectine receptors, Tyrosine kinase, PI3K, glucose transporters, sRAGE and visfatin in insulin resistant/diabetic rats. PLoS One. 2014 Feb 20;9(2):e89169.
114. Kolahdouz Mohammadi R et al. The effect of coenzyme Q10 supplementation on metabolic status of type 2 diabetic patients. Minerva Gastroenterol Dietol. 2013 Jun;59(2):231-6.
115. Hodgson JM et al. Coenzyme Q10 improves blood pressure and glycaemic control: a controlled trial in subjects with type 2 diabetes. Eur J Clin Nutr. 2002 Nov;56(11):1137-42.
116. Hamilton SJ et al. Coenzyme Q10 improves endothelial dysfunction in statin-treated type 2 diabetic patients. Diabetes Care. 2009 May;32(5):810-2.
117. Lindberg S et al. Adiponectin, type 2 diabetes and cardiovascular risk. European Journal of Preventive Cardiology, 21 Nov 2013, 22(3):276-283
118. Yoritaka A et al. Randomized, double-blind, placebo-controlled pilot trial of reduced coenzyme Q10 for Parkinson's disease. Parkinsonism Relat Disord. 2015 Aug;21(8):911-6.
119. Zhang YP et al. Prophylactic and antinociceptive effects of coenzyme Q10 on diabetic neuropathic pain in a mouse model of type 1 diabetes. Anesthesiology. 2013 Apr;118(4):945-54.
120. Langsjoen PH et al. Supplemental ubiquinol in patients with advanced congestive heart failure. Biofactors. 2008;32(1-4):119-28.
121. Hosoe K et al. Study on safety and bioavailability of ubiquinol (Kaneka QH) after single and 4-week multiple oral administration to healthy volunteers. Regul Toxicol Pharmacol. 2007 Feb;47(1):19-28.
122. Alamdari A et al. An inverse association between serum vitamin D levels with the presence and severity of impaired nerve conduction velocity and large fiber peripheral neuropathy in diabetic subjects. Neurol Sci. 2015 Jul;36(7):1121-6.
123. Soderstrom LH et al. Association between vitamin D and diabetic neuropathy in a nationally representative sample: results from 2001-2004 NHANES. Diabet Med. 2012 Jan;29(1):50-5.
124. Esteghamati A et al. Non-linear contribution of serum vitamin D to symptomatic diabetic neuropathy: A case-control study. Diabetes Res Clin Pract. 2016 Jan;111:44-50.
125. Shillo P et al. Reduced vitamin D levels in painful diabetic peripheral neuropathy. Diabet Med. 2019 Jan;36(1):44-51.
126. Lips P et al. Vitamin D and type 2 diabetes. J Steroid Biochem Mol Biol. 2017 Oct;173:280-285.
127. Vondra K et al. Vitamin D and new insights into pathophysiology of type 2 diabetes. Horm Mol Biol Clin Investig. 2021 Mar 1;42(2):203-208.
128. Chatterjee R et al. Effect of intra-trial mean 25(OH)D level on diabetes risk, by race and weight: an ancillary analysis in the D2d randomized study cohort. Am J Clin Nutr. 2023 Jul;118(1):59-67.
129. Mohd Ghozali N et al. Mechanisms Linking Vitamin D Deficiency to Impaired Metabolism: An Overview. Int J Endocrinol. 2022 Jul 6;2022:6453882.
130. Ao T et al. The Effects of Vitamin D on Immune System and Inflammatory Diseases. Biomolecules. Nov 3 2021;11(11)
131. Ghadiri-Anari A et al. Dose vitamin D supplementations improve peripheral diabetic neuropathy? A before-after clinical trial. Diabetes Metab Syndr. 2019 Jan - Feb;13(1):890-3.
132. Shehab D et al. Prospective evaluation of the effect of short-term oral vitamin d supplementation on peripheral neuropathy in type 2 diabetes mellitus. Med Princ Pract. 2015;24(3):250-6.
133. Razzaghi R et al. The effects of vitamin D supplementation on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. J Diabetes Complications. 2017 Apr;31(4):766-72.
134. Yorek MA. Is Fish Oil a Potential Treatment for Diabetic Peripheral Neuropathy? Curr Diabetes Rev. 2018;14(4):339-49.
135. Yorek MA. The Potential Role of Fatty Acids in Treating Diabetic Neuropathy. Curr Diab Rep. 2018 Aug 25;18(10):86.
136. Gerbi A et al. Fish oil supplementation prevents diabetes-induced nerve conduction velocity and neuroanatomical changes in rats. J Nutr. 1999 Jan;129(1):207-13.
137. Li MY et al. Dietary fish oil inhibits mechanical allodynia and thermal hyperalgesia in diabetic rats by blocking nuclear factor-kappaB-mediated inflammatory pathways. J Nutr Biochem. 2015 Nov;26(11):1147-55.
138. Tatsumi Y et al. Omega-3 polyunsaturated fatty acids exert anti-oxidant effects through the nuclear factor (erythroid-derived 2)-related factor 2 pathway in immortalized mouse Schwann cells. J Diabetes Investig. 2019 May;10(3):602-12.
139. Soleimani Z et al. Clinical and metabolic response to flaxseed oil omega-3 fatty acids supplementation in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. J Diabetes Complications. 2017 Sep;31(9):1394-400.
140. Stirban A et al. Effects of n-3 fatty acids on macro- and microvascular function in subjects with type 2 diabetes mellitus. Am J Clin Nutr. 2010 Mar;91(3):808-13.
141. Kim H-J et al. Nicotinamide adenine dinucleotide: An essential factor in preserving hearing in cisplatin-induced ototoxicity. Hear Res. 2015 Aug;326:30-9.
142. Poulose N et al. Sirtuin regulation in aging and injury. Biochim Biophys Acta. 2015 Nov;1852(11):2442-55.
143. Braidy N et al. Age related changes in NAD+ metabolism oxidative stress and Sirt1 activity in wistar rats. PLoS One. 2011 Apr 26;6(4):e19194.
144. Gomes AP et al. Declining NAD(+) induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell. 2013 Dec 19;155(7):1624-38.
145. Yoshino J et al. Nicotinamide mononucleotide, a key NAD(+) intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 2011 Oct 5;14(4):528-36.
146. Canto C et al. NAD(+) Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab. 2015 Jul 7;22(1):31-53.
147. Imai S-I et al. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 2014 Aug;24(8):464-71.
148. Wen C et al. Taurine is Involved in Energy Metabolism in Muscles, Adipose Tissue, and the Liver. Mol Nutr Food Res. Jan 2019;63(2):e1800536.
149. Ito T et al. The potential usefulness of taurine on diabetes mellitus and its complications. Amino Acids. 2012 May;42(5):1529-39.
150. Franconi F et al. Taurine levels in plasma and platelets in insulin-dependent and non-insulin-dependent diabetes mellitus: correlation with platelet aggregation. Adv Exp Med Biol. 1994;359:419-24.
151. Sak D et al. The Relationship between Plasma Taurine Levels and Diabetic Complications in Patients with Type 2 Diabetes Mellitus. Biomolecules. 2019 Mar 11;9(3):96.
152. Tao X et al. The effects of taurine supplementation on diabetes mellitus in humans: A systematic review and meta-analysis. Food Chem (Oxf). Jul 30 2022;4:100106.
153. Masouleh SS et al. The Effects of TRX Suspension Training Combined with Taurine Supplementation on Body Composition, Glycemic and Lipid Markers in Women with Type 2 Diabetes. Nutrients. Nov 5 2021;13(11)
美国糖尿病协会
http://www.diabetes.org
美国梅奥诊所
www.mayoclinic.org
美国国立糖尿病、消化和肾病研究所
http://www.niddk.nih.gov
美国糖尿病资讯交换中心
http://diabetes.niddk.nih.gov
加拿大糖尿病协会
http://www.diabetes.ca
加拿大公众卫生局
http://www.phac-aspc.gc.ca
免责声明和安全信息
英文名称:Diabetes Complications
概述
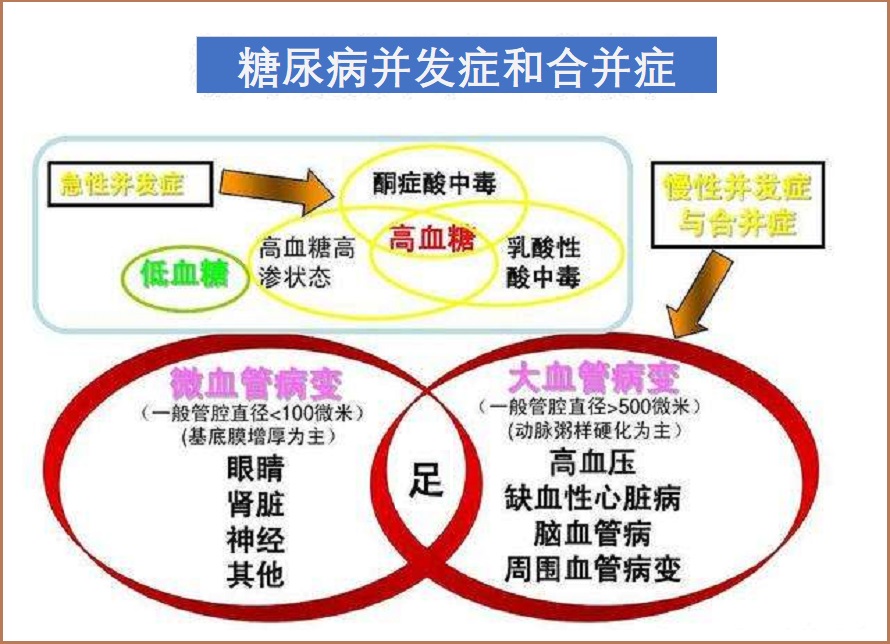
糖尿病是以慢性高血糖为特征的终身代谢性疾病。长期高血糖形成的糖毒性,导致神经和血管损伤病变,累及全身性组织和器官,产生多种并发症而危及糖尿病患者的生命安全。然而,在临床上,治疗糖尿病的手段仅停留在控制血糖方面,对糖尿病及其并发症的病因至今缺乏有效治疗措施。许多研究表明,一些天然化合物可以抑制神经损伤和血管病变,辅助血糖控制治疗,可以降低或延缓糖尿病慢性并发症。并发症病因及机制
糖尿病并发症分为急性和慢性二大类:1.糖尿病急性并发症:包括糖尿病酮症酸中毒、乳酸性中毒和高血糖高渗性昏迷等。发病原因主要是由于胰岛素重度缺乏及升糖激素异常升高,导致血糖过高,引起糖、脂肪和蛋白质代谢紊乱,以致机体水、电解质和酸碱平衡失调等急性发作。此外,在糖尿病降糖治疗过程中突然出现的低血糖昏迷反应。
2.糖尿病慢性并发症:这是糖尿病致残、致死的主要原因,主要类型如下:
- 大血管并发症:心血管、脑血管和下肢血管病变等。
- 微血管并发症,如肾脏病变、视网膜或眼底病变。
- 神经病变,主要是周围神经病变包括感觉神经、运动神经,以及负责内脏器官、内分泌功能的自主神经病变等。
- 糖基化与葡萄糖毒性:高血糖分子可绑定蛋白、脂肪和核酸即糖基化,并形成晚期糖基化终产物(AGEs)而存在于体内多种组织中,尤其是大小血管而导致不可逆性损害,造成血管损伤和神经病变,并逐渐导致多种并发症,包括糖尿病视网膜病、肾病和糖尿病足等。二甲双胍、肌肽、苯磷硫胺和5磷酸吡哆醛(维生素B6衍生物)可遏止糖基化反应及AGEs损伤。
- 低度炎症与葡萄糖毒性:长期高血糖和AGEs积聚可产生多种炎性因子,包括肿瘤坏死因子-α,C-反应蛋白和白细胞介素-6等。
- 增加循环的炎性因子既进一步降低胰岛素敏感性,也损害多种组织,导致多种糖尿病合并症,如高血压、心脑血管病等。
- 这些炎性因子还损害β细胞功能,降低胰岛素分泌。
- 促炎细胞因子对全身产生负面影响。这些细胞因子水平升高会“开启”导致肌肉和骨骼分解和神经元变性、加速损伤 DNA 通路,从而增加死亡风险。
- 自由基与葡萄糖毒性:高血糖和AGEs可导致过量的超氧自由基,日积月累地损伤各种组织,导致多种糖尿病并发症。
糖尿病并发症主要症状
糖尿病基础病理由氧化应激、炎症反应,以及相关的神经病变和血管损伤等组成。因此,糖尿病本质上是一种可涉及到全身性的疾病。据报道,糖尿病并发症可达到100种以上,主要包括如下- 糖尿病肾病:糖尿病患者最重要的合并症之一,发病率呈上升趋势,并构成终末期肾脏病的主因。终末期肾病难以治疗,只能控制血糖、早期干预以延缓发病。
- 糖尿病眼病:
- 糖尿病视网膜病变:这是糖尿病微血管病变最重要的表现,也是糖尿病的最常见的、严重并发症之一。临床上根据是否出现视网膜新生血管为标志,将没有视网膜新生血管形成的糖尿病视网膜病变称为非增殖性糖尿病性视网膜病变;将有视网膜新生血管形成的糖尿病视网膜病变称为增殖性糖尿病视网膜病变。更多相关内容可参阅本网站专文:糖尿病视网膜病变 >>
- 糖尿病白内障:通常发生在血糖控制不良的青少年糖尿病患者(1型糖尿病)。多为双眼发病,发展迅速,甚至可于数天、数周或数月内发展为完全混浊。
- 糖尿病性葡萄膜炎。
- 糖尿病足病:由糖尿病外周血管病变和周围神经病变合并引起。从轻度的神经麻木、刺痛等症状,到严重的足部溃疡、感染后难愈合甚至需要截肢等。更多相关内容可参阅本网站专文:糖尿病足 >>
- 糖尿病合并心脏病:AGEs可引起包括心脏大小血管病变、植物神经病变,涉及到动脉粥样硬化、心肌病变等。此外,高血糖也造成血脂异常,导致高血压合并症,显著增加心血管病风险。研究表明,糖尿病患者冠心病的死亡风险比非糖尿患者群高3~5倍。
- 糖尿病脑血管病:由糖尿病所引起的颅内大血管和微血管病变。主要表现为脑动脉硬化、脑血栓、脑溢血和脑萎缩等。据统计,2型糖尿病患者有20%~40%会发生脑血管病。
- 糖尿病神经病变:主要类型为糖尿病周围神经疾病,即慢性远端对称性、感觉运动性的多发神经病变。其发病率很高,实际上在糖尿病前期就已经存在了。临床上,至今仍然缺乏有效治疗措施,只能对症治疗。因此,重点在于控制血糖、延缓病症发展。更多相关内容可参阅本网站专文:糖尿病神经病变 >>
血糖调节及糖尿病发展
葡萄糖是细胞的主要能源。葡萄糖主要来源于食物中的碳水化合物,而肝脏通过糖异生也制造葡萄糖以维持正常血糖水平。胰岛素促进细胞吸收血液中的葡萄糖。如果胰岛素不足或作用异常,就导致高血糖和糖尿病。影响血糖水平的因素主要如下:- 日常饮食:精制的碳水化合物(如糖、米和面等)对餐后血糖影响最大,其快速分解成葡萄糖进入血液形成餐后高血糖。研究表明,餐后高血糖是心血管病的独立危险因素。
- 超重或肥胖:可损害胰岛素敏感性和葡萄糖利用。
- 脂肪组织可释放干扰胰岛素作用的细胞因子(如肿瘤坏死因子-α等),使巨噬细胞浸润脂肪组织,导致胰岛素抵抗。因此,减轻体重可改善胰岛素敏感性,并减少脂肪组织的炎症基因表达。
- 肥胖会下调脂联素(Adiponectin)表达,因此降低胰岛素敏感性。
- 肥胖引起瘦素(Leptin)抵抗,从而影响热量消耗、增加内脏脂肪累积和促炎反应,加重胰岛素抵抗。
更多相关内容可参阅本网专文:体重管理法 >>
- 肝功能与血糖调节:肝脏是重要的血糖调节器官,通过肝糖原储存和分解、糖异生来维持血糖稳定。
- 糖原分解与胰岛素:在血糖水平不足(如夜晚禁食或剧烈运动后)时,肝糖原被分解进入血液中维持血糖水平。胰岛素可抑制肝糖原分解,如果存在胰岛素抵抗,则肝糖原不受控制地分解葡萄糖,使血糖水平异常。
- 糖异生:是指肝脏将非糖前体(氨基酸、乳酸和甘油等)转变为葡萄糖或糖原的过程,以维持血糖水平。胰岛素抵抗导致糖异生信号紊乱,这是2型糖尿病早上空腹血糖高的原因。二甲双胍可抑制糖异生过度,控制血糖水平。
肝功能异常或慢性肝病则可引起血糖调节紊乱,导致所谓“肝源性糖尿病”。
- 参与血糖调节的主要激素:
- 胰岛素:饭后血糖升高时,刺激胰岛β细胞分泌胰岛素进入血液中,以促进葡萄糖被肌肉、脂肪和肝细胞等所利用并能稳定血糖水平;胰岛素还通过抑制肝糖原分解来防止高血糖。如果存在胰岛素抵抗,则葡萄糖难以通过细胞膜进入细胞被利用,结果血液葡萄糖含量升高。于是又刺激胰腺分泌更多胰岛素,胰岛素抵抗加剧、2型糖尿病就形成了。长期高胰岛素也导致脂肪存储沉积(超重或肥胖)、引起心血管病等代谢性疾病。
- 胰高血糖素:由胰腺的α细胞产生,与胰岛素共同调节血糖水平。当血糖水平不足时,刺激胰腺分泌胰高血糖素促使肝糖原分解成葡萄糖进入血液;如果肝糖原耗尽,则由氨基酸等非碳水化合物中产生葡萄糖(糖异生)进入血液。
- 皮质醇(或应激激素):由肾上腺产生的类固醇激素,在对压力的反应中起关键作用。高皮质醇可刺激糖异生,并能使肌肉和脂肪组织对胰岛素产生抵抗。因此,长期的身心压力可导致胰岛素抵抗和内脏脂肪累积。
- 儿茶酚胺:肾上腺素、去甲肾上腺素和多巴胺是响应身体或情绪压力释放到血液中的激素。除增加心率和血压外,儿茶酚胺还增加血糖水平,为“战斗或逃跑”本能反应提供能量。此外,儿茶酚胺通过刺激肝脏中的糖原分解和糖异生作用以及抑制胰岛素刺激的糖原合成来升高血糖水平。
- 肠促胰岛素(Incretin):由胃肠道细胞分泌的激素,在食物摄入后几分钟内直接进入血液。它们刺激胰岛素分泌。胰高血糖素样肽1(GLP-1)和葡萄糖依赖性促胰岛素多肽(GIP)是非常重要的肠促胰岛素。二者占餐后葡萄糖刺激的胰岛素分泌的70%。此外,GLP-1延迟胃排空、抑制食欲及抑制胰高血糖素分泌并减慢肝脏中的葡萄糖生成。使用GLP-1模拟药物(例如艾塞那肽,利拉鲁肽)的低血糖风险较低,因为它们的作用取决于血流中高水平的葡萄糖。 GLP-1还可以保护产生胰岛素的β细胞免于凋亡并促进其增殖。其他影响肠促胰岛素功能的药物是DPP-IV抑制剂(例如西他列汀Januvia)。该药物阻断了通常迅速降解GLP-1的酶(二肽基肽酶IV)的作用。结果,更多的肠促胰岛素被留在循环中。
- 脂联素和瘦素:这是由脂肪组织细胞分泌的激素。脂联素增加了肌肉和肝细胞中的葡萄糖吸收和脂肪“燃烧”,并降低了肝脏中的糖异生。研究发现,长寿的人体内脂联素浓度较高;瘦素作用于大脑的下丘脑,可控制食欲,并促进脂肪“燃烧”和消耗能量。瘦素抵抗是人体无法充分响应瘦素引起饱腹感,燃烧脂肪的信号的状态。瘦素抵抗是肥胖的主要因素,并阻碍体重减轻。胰岛素抵抗和肥胖个体通常具有升高的瘦素水平,但是对其作用具有抵抗力。炎性标志物C反应蛋白通过与瘦素结合并降低其信号传导能力而加剧瘦素抵抗和体重增加。
营养干预措施
以下是基于循证医学和营养学有关文献综合的结果。在稳定健康的血糖水平上,下列干预路径和措施和有助于抑制糖尿病并发症发生和发展。
1.抑制糖基化终产物(AGEs):
糖基化是葡萄糖和其他糖与蛋白质、脂质和核酸不可逆地结合,导致它们功能失调的过程。糖基化产生的功能失调分子被称为晚期糖基化终产物(AGEs)1,2,3。AGEs通过抑制神经功能(进而影响其活动)和触发炎症反应(更多损害神经细胞)来损伤神经4。
1.1.苯磷硫胺:
糖尿病和肥胖通常会引起硫胺素(维生素B1)相对缺乏,这会导致高血糖的破坏性作用5,6。苯磷硫胺是硫胺素(维生素B1)的脂溶性衍生物,其生物利用度比硫胺素高得多,并且能够在血液中达到口服硫胺素浓度的数倍7。这种独特形式的维生素B1可抑制AGEs的形成、炎症和氧化应8,9,6。
一项针对165名糖尿病神经病变患者的临床试验发现,连续六周补充苯磷硫胺可以减轻糖尿病神经病变的疼痛。与每天服用300mg的受试者相比,每天服用600mg苯磷硫胺的益处更为明显。此外,服用苯磷硫胺的受试者益处更长久10,11。
在一项针对13名2型糖尿病受试者的临床试验中,参与者在每天1050mg的苯磷硫胺3天疗程前后吃了一顿高AGEs餐,在两次高AGEs餐后评估受试者的血管和内皮功能。苯磷硫胺给药完全预防了血管功能障碍的迹象,内皮功能障碍和氧化应激的生物标志物显著减少12。
临床和动物研究已经证明了苯磷硫胺在治疗糖尿病相关神经病变、肾病、外周血管疾病和视网膜病变方面的疗效13-15。
1.2.肌肽:
肌肽能够抑制AGEs的形成,甚至逆转蛋白质糖基化16。在一项针对糖尿病小鼠的研究中,补充肌肽使血浆肌肽水平增加了20倍,甘油三酯水平降低了23%,并提高了动脉粥样硬化病变的稳定性17。肌肽还被证明可以提高细胞在高浓度葡萄糖存在下的生存能力,并改善糖尿病动物的伤口愈合18。糖尿病动物模型显示,补充肌肽可以提高红细胞改变其形状的能力,这是血液流动过程中遇到的机械力所必需的;这一过程在糖尿病中受损,导致糖尿病并发症19。
1.3.维生素B6(吡哆醛):
维生素B6,特别其活性形式5磷酸吡哆醛(PLP,简称吡哆醛)参与葡萄糖代谢的几个方面,是一种有效的抗糖化剂20。除了防止蛋白质糖基化,PLP还是脂质(脂肪)糖基化的有效抑制剂21。与健康对照组相比,糖尿病患者的脂质AGEs升高,脂质AGEs的积累可能导致与糖尿病相关的血管疾病22。AGEs的高膳食摄入也会增加糖尿病患者的心血管疾病风险。
较低的血清PLP水平与糖尿病的发病率和进展有关,维生素B6的抗糖化特性可能有助于预防糖尿病并发症20。研究表明,在日本2型糖尿病患者中,维生素B6的摄入量越高,糖尿病视网膜病变的发生率越低23。
对人类和动物的干预研究也很有前景。例如,用35mg PLP、3mg活性叶酸和2mg维生素B12治疗20例2型糖尿病患者,可改善糖尿病周围神经病变的皮肤感觉24。在一项随机对照试验中,与安慰剂相比,44名肥胖或超重女性接受80mg盐酸吡哆醇(维生素B6的主要形式)治疗8周,胰岛素敏感性提高,脂肪量减少25。PLP的补充显著降低了糖尿病大鼠中高浓度的糖基化诱导的毒性化合物,并阻止了糖尿病肾病的进展21,26。
最近的一项研究进一步确定,接受PLP治疗的糖尿病大鼠与糖尿病相关的问题有所减少,包括氧化应激参数下降、血糖水平下降以及肝脏和肾脏损伤恢复27。此外,吡哆胺(Pyridoxamine维生素B6的另一种形式)改善了肥胖2型糖尿病小鼠的胰岛素敏感性28。已知吡哆胺可抑制AGEs和活性氧形成等29,30。
2.抑制神经病变:
2.1.硫辛酸:
它兼具水溶性和脂溶性,可在体内产生并发挥多种生物学作用,尤其是在葡萄糖代谢方面31。事实上,硫辛酸在德国已被批准用于治疗糖尿病神经病变31,32。此外,硫辛酸可能对糖尿病患者的葡萄糖代谢产生积极影响33。硫辛酸帮助对抗糖尿病神经病变的重要机制包括抑制糖化和炎症34,45。据报道,硫辛酸可以在神经元细胞培养中防止氧化损伤36。它还改善了神经的血液流动,使它们能够更有效地利用能量31,37。
一项临床试验表明,在3周内每天3次服用600mg的α-硫辛酸,可显著改善12名糖尿病神经病变患者的症状。在另一项试验中,181名患者被分为每天接受安慰剂或600、1200或1800mg α-硫辛酸组。1800mg/天组在短短一周内症状显著改善,600mg和1200mg组在第2周时症状有所改善31。在这两项试验中,疼痛和灼烧感都得到了缓解。另一项研究发现,在4年的时间里,每天口服600mg硫辛酸可以改善神经性症状,减缓轻度至中度糖尿病神经病变的进展,阻止其进一步恶化38。总之,口服硫辛酸可缓解疼痛、灼烧麻木感等,减少肌无力,提高整体生活质量39,40。
其他试验表明,静脉注射硫辛酸对糖尿病神经病变也有益。一项研究表明,每天静脉注射α-硫辛酸3周是治疗糖尿病神经病变的有效方法,另一项试验报告称,静脉注射2-4周是有效的31,41。
注意:硫辛酸有二种形式的产品:α-硫辛酸(含s-和r-型硫辛酸各占50%)和r-硫辛酸。有证据表明,r-硫辛酸的钠盐比a-硫辛酸更具生物利用性42。
2.2.乙酰L肉碱(ALC)和L肉碱:
肉碱是一种对线粒体能量产生很重要的氨基酸类化合物。对肉碱在糖尿病神经病变中的作用源于有证据表明,患有并发症(包括糖尿病神经病变)的糖尿病患者的血清和总肉碱水平低于没有并发症的糖尿病患者43。因此,ALC和L-肉碱均已被研究为糖尿病神经病变的潜在治疗方法。补充肉碱可能有助于对抗肉碱缺乏症,改善胰岛素抵抗,使细胞更有效地利用葡萄糖,帮助受损神经纤维再生,或帮助受损神经元更有效地运输细胞内成分43。
已经对补充肉碱对糖尿病神经病变的影响进行了多项人体试验。一项研究发现,L-肉碱(每天2g,持续10个月)可以改善神经传导速度,而神经传导速度在糖尿病神经病变中受损44。对ALC的研究发现,它减轻了糖尿病神经病变患者的疼痛,改善了腿部的振动感,并增加了神经再生45-47。补充肉碱也可能有助于治疗糖尿病引起的自主神经病变,对动物模型进行的一项研究发现ALC降低了糖尿病自主神经病变的心血管体征48。
ALC对外周神经系统具有神经保护和镇痛作用49;可防止神经细胞死亡、促进神经再生和神经损伤修复以及增强对神经生长因子的反应等50。在糖尿病患者中,剂量为1500mg/天至3000mg/天的乙酰-L-肉碱可提高神经传导速度和强度,降低疼痛和残疾评分,增加神经纤维数量,并再生受损的神经纤维51,52,46。
2.3.十六酰胺乙醇(PEA):
一种天然存在于全身组织中的脂质化合物,包括中枢神经系统。在大豆卵磷脂、蛋黄和花生等食物中也存在53。越来越多的研究表明,补充PEA可能有效缓解各种原因引起的疼痛,而不会引发不良副作用54,55。大多数现有的临床前研究表明,PEA通过改变某些基因的表达和减少炎症信号传导发挥作用,但也提出了其镇痛作用的其他可能机制,包括其通过神经系统中的大麻素受体刺激信号传导的能力56-58。
几项临床试验表明,PEA可以减轻多种原因引起的疼痛,包括糖尿病神经病变、化疗诱导的周围神经病变、坐骨神经压迫、腕管综合征、骨关节炎、腰痛、背部手术失败、中风相关神经痛、多发性硬化症、牙痛、慢性骨盆疼痛、疱疹后神经痛和阴道疼痛等59。在一项针对因各种情况而无法通过常规治疗控制疼痛的慢性疼痛患者的观察性研究中,在三周内每天二次添加600mg PEA,然后在四周内每天一次,降低了所有完成研究的参与者的平均疼痛强度得分60。
在一项随机对照试验中,636名因坐骨神经压迫而疼痛的参与者接受了每天300mg PEA、每天600mg PEA或安慰剂以及他们常用的止痛药,为期三周。两种剂量的PEA都比安慰剂更能减轻疼痛,而且高剂量比低剂量更有效。事实上,600mg组的疼痛评分降低了50%以上61。在118名神经疼痛患者中,30天的标准治疗加上每天600mg PEA比单独的标准治疗更有效62。一项随机临床试验发现,在缓解颞下颌关节(TMJ)疼痛方面,一周内每天900mg PEA,一周后每天600mg PEA比布洛芬更有效,布洛芬的剂量为600mg,两周内每天三次63。
对PEA的微粉化制剂也进行了研究。微粉化会产生更小的颗粒,这些颗粒可能更容易被吸收。剂量为600–1200mg/天的微粉化PEA可减轻糖尿病或创伤相关神经疼痛、背部手术失败后的慢性疼痛和拔牙后的急性疼痛受试者的疼痛64-66。在一份关于100例与脊柱疾病相关的神经疼痛的报告中,将超微粉化PEA补充剂纳入疼痛管理治疗显示出了有希望的结果67。一项荟萃分析发现,患有子宫内膜异位症引起的慢性盆腔疼痛的女性似乎受益于每天800mg微粉化PEA加上每天80mg虎杖苷(Polydatin)的组合68。在一项随机对照试验中,PEA和虎杖苷的组合在减轻肠易激综合征患者腹痛方面比安慰剂更有效69。
2.4. N-乙酰半胱氨酸(NAC):
既是体内谷胱甘肽合成前体,NAC本身也是一种抗氧化剂。它可以保护神经元免受氧化损伤70。此外,NAC已被证明可以抑制AGEs的形成71。对糖尿病神经病变动物模型的多项研究发现,NAC可以防止神经元死亡并防止神经损伤70,32。
2.5.维生素B族:
B族维生素是一种维生素家族,在人体中发挥着许多作用,尤其是在细胞能量生成和神经系统功能中72。
- 硫胺素(B1)和苯磷硫胺:硫胺素缺乏可直接导致神经病变32。先前研究发现硫胺素可以用于治疗疼痛的糖尿病神经病变。苯磷硫胺是硫胺素的脂溶性衍生物,更容易被消化道吸收73。苯磷硫胺可能调节导致糖尿病神经病变的几种途径:AGEs的形成、蛋白激酶C途径,以及由于高糖水平可能在细胞内发生的损伤性变化74,6。
- 维生素B12:它对神经系统的功能至关重要,缺乏B12会导致严重的周围神经病变32。此外,糖尿病神经病变患者通常具有高水平的破坏血管的化合物—同型半胱氨酸,在维生素B12水平较低的情况下,同型半胱氨酸会升高75。还研究了补充维生素B12治疗糖尿病神经病变的潜在益处,维生素B12的活性形式—甲钴胺,它对神经组织具有亲和力,在这方面已经得到了广泛的研究76。对糖尿病神经病变动物模型的研究发现,甲钴胺可以减轻糖尿病神经病变引起的损害,可能通过调节蛋白激酶C信号通路或激活化学信号来帮助神经存活和再生76,77。
在多项临床试验中发现,2mg甲钴胺、3mg L-甲基叶酸和35mg 5’-磷酸吡哆醛(维生素B6形式之一)的组合可以改善神经病变症状,并有助于维持下肢小神经的健康78,79。这三种营养素的组合也被发现有助于降低糖尿病神经病变患者的住院和医疗费用80。对甲钴胺的单独研究也令人鼓舞,口服甲钴胺(每天1500mcg)和注射甲钴胺(每天2000mcg)均可改善麻木、反射、对振动的敏感性、针刺样刺激、步态和疼痛81,82。一项研究甚至发现,甲钴胺比常用于治疗糖尿病神经性疼痛的抗抑郁药去甲替林更有效81。
- 叶酸和维生素B6:与甲钴胺一样,维生素B6对神经功能很重要,而叶酸可能有助于改善供应神经的血管功能79。如前所述,叶酸和维生素B6衍生物以及甲钴胺已在临床试验中进行了测试,并显示出阳性结果78,79。
叶酸还可以降低同型半胱氨酸的水平83,后者是心血管病独立风险因素,而且对神经细胞存在毒性84。在2001年一项涉及65名II型糖尿病患者的研究中,同型半胱氨酸每增加5 mmol/L,神经损伤的风险就会增加一倍多85。患有II型糖尿病并伴有神经病变的中国患者的叶酸水平也明显低于无神经病变的患者86。
最近一项针对糖尿病神经病变患者的研究表明,连续16周每天服用1000mcg叶酸,可降低同型半胱氨酸水平,并显著提高神经传导速度和信号强度87。
2.6.姜黄素:
姜黄素因其抗炎特性而著称,最近也因其在疼痛控制方面的潜力而受到研究88,89。事实上,姜黄素已被证明可以显著提高实验动物的疼痛阈值并降低疼痛超敏反应90-92。
对于患有糖尿病神经损伤的人来说,姜黄素提供的不仅仅是止痛,也可能减缓或逆转最初产生神经性疼痛的一些过程。临床前研究表明,姜黄素可减少TNF-α的产生,TNF-α是一种炎症蛋白,也是疼痛的诱因91,激活内部疼痛缓解系统(称为内源性阿片系统)89;减少细胞中的氧化应激,这是神经疼痛的主要诱因92,以及减少糖尿病神经中的异常电脉冲93,94。
其他实验研究和人体试验表明,姜黄素是一种很有前途的预防和治疗糖尿病及其并发症的天然药物,主要作用机制是中和反应性自由基和减少炎症的能力。姜黄素似乎可以提高胰岛素敏感性,降低血液中的葡萄糖和脂质水平。它还可以保护胰腺中产生胰岛素的β细胞95-97。
3.降低血管损伤、改善内皮功能:
3.1.碧萝芷:
在碧萝芷与代谢综合征(包括高血压、糖尿病等)的综述中发现,它有助于改善血糖指标、降低血压值等98。碧萝芷已被研究用于治疗糖尿病视网膜病变99,它可以保护视网膜中的毛细血管免受损伤。
一项研究77名2型糖尿病患者的临床试验检测了碧萝芷对血管损伤标志物内皮素-1水平的影响,内皮素-1是血管损伤的标志。34名受试者在12周内每天服用100mg碧萝芷,与43名服用安慰剂的受试者相比,内皮素-1水平降低100。一项对1289人进行的多项研究的全面综述发现,碧萝芷对治疗糖尿病视网膜病变可能有用。这篇文章的结论是,60至150mg的碧萝芷剂量有助于减缓糖尿病视网膜病变的进展,提高2型糖尿病患者的视力,提高毛细血管的强度,并减少毛细血管渗漏到视网膜中101。
另一项研究考察了在二个月内每天给2型糖尿病患者服用150mg碧萝芷的效果,发现24名接受碧萝芷治疗的受试者与接受安慰剂治疗的22名受试者相比,视力提高,视网膜肿胀和增厚减少102。除了含有可以中和糖尿病引起的视网膜损伤的化合物外,碧萝芷中的一些化合物可能有助于修复受损的毛细血管99。
2型糖尿病患者合并有相当大的心血管疾病(CVD)的过度发病和死亡风险。一项金标准临床试验(RCT)包括48名受试者,旨在测试碧萝芷在减少2型糖尿病患者降压药物使用和心血管疾病危险因素方面的临床有效性。补充碧萝芷12周后,与对照组相比,碧萝芷改善了糖尿病控制,降低了心血管疾病风险因素,并减少了抗高血压药物的使用103。
3.2.维生素E(γ-生育酚):
餐后血糖飙升会损伤血管内壁,导致内皮功能障碍和血管疾病。γ-生育酚是维生素E的一种形式,具有抗炎和清除自由基的活性。在健康男性中进行的两项试验发现,补充γ-生育酚可以防止餐后葡萄糖峰值诱导的内皮功能障碍相关变化104,105。
3.3葡萄籽提取物:
所含原花青素可调节氧化应激,并已被研究用于多种健康状况,包括高血压、癌症和阿尔茨海默病106-108。葡萄多酚似乎具有重要的抗糖尿病作用,并保护组织免受血糖升高的损害。
在一项针对32名2型糖尿病患者的为期四周的随机安慰剂对照试验中,与安慰剂相比,每天摄入600mg葡萄籽提取物可显著降低果糖胺(Fructosamine)。果糖胺是一种类似于HbA1C的测试,可以测量几周内的血糖水平。葡萄籽提取物还降低了总胆固醇和Hs-CRP,并显著提高了血液谷胱甘肽-人体主要的内源性抗氧化剂之一109。另一项试验发现,红酒中富含葡萄多酚的非酒精成分可以提高胰岛素敏感性,降低心血管风险110。
一项随机对照试验,每天给2型糖尿病的健康但超重的一级亲属服用2g葡萄多酚,为期8周。然后,受试者被大量摄入果糖(果汁和许多加糖饮料中的糖)。在安慰剂组中,果糖激发导致氧化应激增加,胰岛素敏感性和线粒体活性降低。多酚组预防了所有这些负面影响111。
4.维持基础营养:
4.1.辅酶Q10(CoQ10):
CoQ10对线粒体能量代谢至关重要,也是氧化应激的强大抑制剂112。辅酶Q10缺乏与糖尿病有关113,114。在一项针对64名2型糖尿病患者的随机对照试验中,连续12周每天补充200mg辅酶Q10可降低血清HbA1C浓度,并降低总胆固醇和LDL胆固醇水平114。一项针对74名2型糖尿病受试者的临床试验发现,每天两次100mg辅酶Q10可显著降低HbA1C和血压值115。在一项针对23名他汀类药物治疗的2型糖尿病患者的安慰剂对照试验中,每天200mg辅酶Q10显著改善了糖尿病患者血管内皮功能障碍的标志物116。
在糖尿病动物模型中,辅酶Q10治疗显著改善了胰岛素抵抗,降低了血清胰岛素和葡萄糖水平,并将能量调节激素脂联素水平提高了六倍113。高水平的脂联素与糖尿病和心血管并发症的风险降低有关117,118。
动物研究表明,长期使用辅酶Q10对进行性糖尿病神经病变具有保护作用。辅酶Q10的有益作用可能归因于减少氧化损伤和炎症,这两个因素都与糖尿病神经病变有关119。
还原形式的辅酶Q10泛醇比泛醌形式更有效地被吸收,对中老年人尤其适应120,121。
4.2.维生素D:
许多研究证实,维生素D缺乏(低于20ng/mL)和不足(20ng/mL至30ng/mL)与与糖尿病患者的神经病变密切相关122-125。维生素D状态已被证明与葡萄糖代谢有关126,127。在不同人群中,较高水平的血清维生素D被证明可以降低糖尿病风险128。
慢性全身炎症是胰岛素抵抗和糖尿病的一个特征,可能是一种影响,但有人认为它实际上可能是病因127,129。负责炎症的免疫细胞表达维生素D受体,维生素D在多种情况下成功抑制慢性炎症,而维生素D缺乏与促炎状态有关129,130。
一项研究发现,维生素D水平不足的糖尿病患者患神经性疾病的几率高出2倍124。临床试验已经证实,每周50,000IU的维生素D3可以显著减轻糖尿病神经损伤的症状,提高生活质量131,132。在一项研究中,每二周服用50,000IU的维生素D,12周后糖尿病足溃疡的面积显著缩小133。
一般认为,维持血清25-羟基维生素D水平的最佳目标范围在50至80ng/mL之间。
4.3.欧米伽3脂肪酸:
鱼油和亚麻籽油富含抗炎的ω-3脂肪酸。增加ω-3脂肪酸摄入在预防和缓解糖尿病神经病变方面显示出巨大的前景。在啮齿类动物研究中,鱼油可以减缓甚至逆转糖尿病神经损伤的进展134,135。
研究表明,ω-3脂肪酸可能有助于治疗糖尿病神经病变路径包括:糖尿病神经病变患者的神经传导速度下降,神经传导速度衡量电脉冲在神经中的移动速度。动物研究发现,鱼油能迅速恢复神经传导速度,减少对关键神经束的可见损伤136;使用鱼油治疗后,糖尿病动物的疼痛和对触摸的超敏反应减轻,动物的神经簇显示炎症减少,核因子κB(NF-kB,主要促炎因子)水平降低137;在用DHA或EPA处理后,实验室培养的神经细胞会急剧增加其对抗有害氧化应激的蛋白质产量138。
上述不同的作用会产生重大后果。在一项针对糖尿病足溃疡(可导致截肢的神经病变并发症)患者的研究中,每天服用1g ω-3可显著缩小溃疡面积。与此同时,炎症标志物降低,总抗氧化水平显著升高139。
此外,鱼油中的DHA和EPA似乎可以防止与餐后血糖飙升相关的一些血管功能变化。一项针对34名2型糖尿病受试者的为期六周的试验发现,补充ω-3脂肪酸可以显著防止餐后小血管和大血管功能障碍140。
4.4.烟酰胺腺嘌呤二核苷酸(NAD+):
烟酰胺腺嘌呤二核苷酸(NAD+)是细胞能量的关键调节因子141。它也是Sirtuin蛋白的辅因子,该蛋白参与许多代谢活动,并与寿命有关。衰老与SIRT1活性下降有关,SIRT1是编码Sirtuin 1蛋白的基因。临床前研究表明,SIRT1表达增加可延长寿命142。与年龄相关的NAD+水平下降与SIRT1活性降低有关143,144。NAD+代谢也与糖尿病的病因和并发症有关145-147。
众所周知,烟酰胺核糖苷和NMN(β-烟酰胺单核苷酸)都是维生素B3衍生物,补充可以提高细胞NAD+水平。
4.5.牛磺酸:
牛磺酸是一种遍布全身的氨基磺酸,且在大脑、心脏和骨骼肌中含量特别丰富。牛磺酸参与多种重要的生理功能,如促进细胞膜稳定和促进健康的神经系统功能148。临床前研究发现,牛磺酸可能会抵消一些导致糖尿病并发症的问题(如糖尿病肾病、糖尿病白内障和糖尿病心肌病)149。观察性研究发现,与非糖尿病对照组相比,糖尿病患者的血浆牛磺酸浓度往往较低150,151。
一项荟萃分析评估了五项随机对照试验中补充牛磺酸对血糖标志物的影响,该试验共招募了209名参与者。在这些研究中,糖尿病患者每天接受500-3000mg牛磺酸或安慰剂治疗,持续2-16周。与对照组相比,那些服用牛磺酸的人降低了HbA1C和空腹血糖,并在胰岛素抵抗模型中有所改善152。
在荟萃分析中的一项研究中,当牛磺酸与全身阻力运动暂停训练的独特力量训练相结合时,患有2型糖尿病的女性的改善最大。这种训练形式允许使用体重和重力作为阻力进行单关节和多关节训练。当每天两次(早、晚)服用500mg牛磺酸,持续八周,并与此训练相结合时,与只服用一种或另一种或安慰剂的组相比,参与者的体脂百分比和胰岛素抵抗降低幅度更大,高密度脂蛋白增加幅度更大153。这些发现表明,补充牛磺酸与力量训练相结合可能是支持糖尿病患者代谢健康的一种很有前途的干预措施。
综合干预方案
下列是基于循证医学和营养学有关文献综合的干预方案:- 糖尿病并发症管理要略:
- 糖尿病合并症防控:
以及可参阅本网站如下专文的相关内容:
参考文献:
1. Ahmed N et al. Advanced glycation endproducts: what is their relevance to diabetic complications? Diabetes Obes Metab. 2007 May;9(3):233-45.
2. Sugimoto K et al. Role of advanced glycation end products in diabetic neuropathy. Curr Pharm Des. 2008;14(10):953-61.
3. Fournet M et al. Glycation Damage: A Possible Hub for Major Pathophysiological Disorders and Aging. Aging Dis. 2018 Oct;9(5):880-900.
4. Vincent AM et al. Diabetic neuropathy: cellular mechanisms as therapeutic targets. Nat Rev Neurol. 2011 Sep 13;7(10):573-83.
5. Beltramo E et al. Effects of thiamine and benfotiamine on intracellular glucose metabolism and relevance in the prevention of diabetic complications. Acta Diabetol. 2008 Sep;45(3):131-41.
6. Balakumar P et al. The multifaceted therapeutic potential of benfotiamine. Pharmacol Res. 2010 Jun;61(6):482-8.
7. Xie F et al. Pharmacokinetic study of benfotiamine and the bioavailability assessment compared to thiamine hydrochloride. J Clin Pharmacol. 2014 Jun;54(6):688-95.
8. Hammes H-A et al. Benfotiamine blocks three major pathways of hyperglycemic damage and prevents experimental diabetic retinopathy. Nat Med. 2003 Mar;9(3):294-9.
9. Shoeb M et al. Anti-inflammatory effects of benfotiamine are mediated through the regulation of the arachidonic acid pathway in macrophages. Free Radic Biol Med. 2012 Jan 1;52(1):182-90.
10. Stracke H et al. Benfotiamine in diabetic polyneuropathy (BENDIP): results of a randomized, double blind, placebo-controlled clinical study. Exp Clin Endocrinol Diabetes. 2008 Nov;116(10):600-5.
11. Winkler G, Pal B, Nagybeganyi E, et al. Effectiveness of different benfotiamine dosage regimens in the treatment of painful diabetic neuropathy. Arzneimittelforschung. 1999 Mar;49(3):220-4.
12. Stirban A et al. Benfotiamine prevents macro- and microvascular endothelial dysfunction and oxidative stress following a meal rich in advanced glycation end products in individuals with type 2 diabetes. Diabetes Care. 2006 Sep;29(9):2064-71.
13. Nikolic A et al. [The effect of benfothiamine in the therapy of diabetic polyneuropathy]. Srp Arh Celok Lek. 2009 Nov-Dec;137(11-12):594-600.
14. Chakrabarti R et al. Preventive effects of benfotiamine in chronic diabetic complications. J Diabetes Investig. 2011 Apr 7;2(2):123-31.
15. Haupt E et al. Benfotiamine in the treatment of diabetic polyneuropathy--a three-week randomized, controlled pilot study (BEDIP study). Int J Clin Pharmacol Ther. 2005 Feb;43(2):71-7.
16. Boldyrev AA et al. Carnosine as a natural antioxidant and geroprotector: from molecular mechanisms to clinical trials. Rejuvenation Res. 2010 Apr-Jun;13(2-3):156-8.
17. Brown BE et al. Supplementation with carnosine decreases plasma triglycerides and modulates atherosclerotic plaque composition in diabetic apo E(-/-) mice. Atherosclerosis. 2014 Feb;232(2):403-9.
18. Ansurudeen I et al. Carnosine enhances diabetic wound healing in the db/db mouse model of type 2 diabetes. Amino Acids. 2012 Jul;43(1):127-34.
19. Yapislar H et al. Effect of carnosine on erythrocyte deformability in diabetic rats. Arch Physiol Biochem. 2012 Dec;118(5):265-72.
20. Mascolo E et al. Vitamin B6 and Diabetes: Relationship and Molecular Mechanisms. Int J Mol Sci. 2020 May 23;21(10):3669.
21. Higuchi O et al. Aminophospholipid glycation and its inhibitor screening system: a new role of pyridoxal 5'-phosphate as the inhibitor. J Lipid Res. 2006 May;47(5):964-74.
22. Fishman SL et al. The role of advanced glycation end-products in the development of coronary artery disease in patients with and without diabetes mellitus: a review. Mol Med. 2018 Nov 23;24(1):59.
23. Horikawa C et al. Vitamin B6 intake and incidence of diabetic retinopathy in Japanese patients with type 2 diabetes: analysis of data from the Japan Diabetes Complications Study (JDCS). Eur J Nutr. 2020 Jun;59(4):1585-1594.
24. Walker KZ et al. Diet and exercise in the prevention of diabetes. J Hum Nutr Diet. 2010 Aug;23(4):344-52.
25. Haidari E et al. The Effect of Pyridoxine Hydrochloride Supplementation on Leptin, Adiponectin, Glycemic Indices, and Anthropometric Indices in Obese and Overweight Women. Clin Nutr Res. 2021 Jul 22;10(3):230-242.
26. Nakamura S et al. Pyridoxal phosphate prevents progression of diabetic nephropathy. Nephrol Dial Transplant. 2007 Aug;22(8):2165-74.
27. Abdullah KM et al. Anti-diabetic study of vitamin B6 on hyperglycaemia induced protein carbonylation, DNA damage and ROS production in alloxan induced diabetic rats. Toxicol Res (Camb). 2019 Jun 11;8(4):568-579.
28. Mastrocola R et al. Altered hepatic sphingolipid metabolism in insulin resistant mice: Role of advanced glycation endproducts. Free Radic Biol Med. 2021 Jun:169:425-435.
29. Abdullah KM et al. Study of pyridoxamine against glycation and reactive oxygen species production in human serum albumin as model protein: An in vitro & ex vivo approach. Int J Biol Macromol. 2018 Dec;120(Pt B):1734-1743.
30. Ramis R et al. How Does Pyridoxamine Inhibit the Formation of Advanced Glycation End Products? The Role of Its Primary Antioxidant Activity. Antioxidants (Basel). 2019 Sep 1;8(9):344.
31. McIlduff CE et al. Critical Appraisal of the Use of Alpha Lipoic Acid (Thiooctic Acid) in the Treatment of Symptomatic Diabetic Polyneuropathy. Therapeutics and Clinical Risk Management, 2011; 7: 377-385.
32. Head KA. Peripheral Neuropathy: Pathogenic Mechanisms and Alternative Therapies. Alternative Medicine Review, 2006; 11(4): 294-329.
33. Korotchkina LG et al. R-lipoic acid inhibits mammalian pyruvate dehydrogenase kinase. Free radical research. Oct 2004;38(10):1083-1092.
34. Thirunavukkarasu V et al. Lipoic acid improves glucose utilisation and prevents protein glycation and AGE formation. Die Pharmazie. Oct 2005;60(10):772-775.
35. Kunt T et al. Alpha-lipoic acid reduces expression of vascular cell adhesion molecule-1 and endothelial adhesion of human monocytes after stimulation with advanced glycation end products. Clinical science. Jan 1999;96(1):75-82.
36. Bharat S et al. Pre-treatment with R-lipoic acid alleviates the effects of GSH depletion in PC12 cells: implications for Parkinson's disease therapy. Neurotoxicology. Oct 2002;23(4-5):479-486.
37. Bertolotto F et al. Combination of Alpha Lipoic Acid and Superoxide Dismutase Leads to Physiological and Symptomatic Improvements in Diabetic Neuropathy. Drugs Research and Development, 2012; 12(1): 29-34.
38. Ziegler D et al. Efficacy and Safety of Antioxidant Treatment with Alpha-Lipoic Acid Over 4 Years in Diabetic Polyneuropathy. Diabetes Care, 2011; 34: 2054-2060.
39. Garcia-Alcala H et al. Treatment with alpha-Lipoic Acid over 16 Weeks in Type 2 Diabetic Patients with Symptomatic Polyneuropathy Who Responded to Initial 4-Week High-Dose Loading. J Diabetes Res. 2015;2015:189857.
40. Agathos E et al. Effect of alpha-lipoic acid on symptoms and quality of life in patients with painful diabetic neuropathy. J Int Med Res. 2018 May;46(5):1779-90.
41. Han T et al. A Systematic Review and Meta-Analysis of Alpha-Lipoic Acid in the Treatment of Diabetic Peripheral Neuropathy. European Journal of Endocrinology, 2012; 167: 465-471.
42. Carlson DA et al. The plasma pharmacokinetics of R-(+)-lipoic acid administered as sodium R-(+)-lipoate to healthy human subjects. Alternative medicine review. Dec 2007;12(4):343-351.
43. Evans JD et al. Role of Acetyl-L-Carnitine in the Treatment of Diabetic Peripheral Neuropathy. Annals of Pharmacotherapy, 2008; 42: 1686-1691.
44. Ulvi H et al. Effect of L-Carnitine on Diabetic Neuropathy and Ventricular Dispersion in Patients with Diabetes Mellitus. Turkish Journal of Medical Science, 2010; 40(2): 169-175.
45. Bansal V et al. Diabetic Neuropathy. Postgraduate Medical Journal, 2006; 82: 95-100.
46. Sima AA et al. Acetyl-L-Carnitine Improves Pain, Nerve Regeneration, and Vibratory Perception in Patients with Chronic Diabetic Neuropathy. Diabetes Care, 2005; 28(1): 89-94.
47. Adriaensen H et al. Critical Review of Oral Drug Treatments for Diabetic Neuropathic Pain – Clinical Outcomes Based on Efficacy and Safety Data from Placebo-Controlled and Direct Comparative Studies. Diabetes/Metabolism Research and Reviews, 2005; 21: 231-240.
48. Giudice PL et al. Autonomic Neuropathy in Streptozotocin Diabetic Rats: Effect of Acetyl-L-Carnitine. Diabetes Research and Clinical Practice, 2002; 56: 173-180.
49. Onofrj M et al. Acetyl-L-carnitine: from a biological curiosity to a drug for the peripheral nervous system and beyond. Expert Rev Neurother. 2013 Aug;13(8):925-36.
50. Sergi G et al. Effects of acetyl-L-carnitine in diabetic neuropathy and other geriatric disorders. Aging Clin Exp Res. 2018 Feb;30(2):133-8.
51. De Grandis D et al. Acetyl-L-carnitine (levacecarnine) in the treatment of diabetic neuropathy. A long-term, randomised, double-blind, placebo-controlled study. Drugs R D. 2002;3(4):223-31.
52. Li S et al. Effects of acetyl-L-carnitine and methylcobalamin for diabetic peripheral neuropathy: A multicenter, randomized, double-blind, controlled trial. J Diabetes Investig. 2016 Sep;7(5):777-85.
53. Mattace Raso G et al. Palmitoylethanolamide in CNS health and disease. Pharmacological Research. Aug 2014;86:32-41.
54. Artukoglu BB et al. Efficacy of Palmitoylethanolamide for Pain: A Meta-Analysis. Pain Physician. Jul 2017;20(5):353-362.
55. Paladini A et al. Palmitoylethanolamide, a Special Food for Medical Purposes, in the Treatment of Chronic Pain: A Pooled Data Meta-analysis. Pain Physician. Feb 2016;19(2):11-24.
56. Gabrielsson L et al. Palmitoylethanolamide for the treatment of pain: pharmacokinetics, safety and efficacy. British Journal of Clinical Pharmacology. Oct 2016;82(4):932-942.
57. Khasabova IA et al. Peroxisome proliferator-activated receptor alpha mediates acute effects of palmitoylethanolamide on sensory neurons. The Journal of Neuroscience. Sep 12 2012;32(37):12735-12743.
58. Di Cesare Mannelli L et al. Palmitoylethanolamide is a disease-modifying agent in peripheral neuropathy: pain relief and neuroprotection share a PPAR-alpha-mediated mechanism. Mediators of Inflammation. 2013;2013:328797.
59. Keppel Hesselink JM et al. Therapeutic utility of palmitoylethanolamide in the treatment of neuropathic pain associated with various pathological conditions: a case series. Journal of Pain Research. 2012;5:437-442.
60. Gatti A et al. Palmitoylethanolamide in the treatment of chronic pain caused by different etiopathogenesis. Pain Medicine (Malden, Mass.). Sep 2012;13(9):1121-1130.
61. Keppel Hesselink JM et al. Palmitoylethanolamide, a neutraceutical, in nerve compression syndromes: efficacy and safety in sciatic pain and carpal tunnel syndrome. Journal of Pain Research. 2015;8:729-734.
62. Dominguez CM et al. N-palmitoylethanolamide in the treatment of neuropathic pain associated with lumbosciatica. Pain Management. Mar 2012;2(2):119-124.
63. Marini I et al. Palmitoylethanolamide versus a nonsteroidal anti-inflammatory drug in the treatment of temporomandibular joint inflammatory pain. Journal of Orofacial Pain. Spring 2012;26(2):99-104.
64. Cocito D et al. Short-term efficacy of ultramicronized palmitoylethanolamide in peripheral neuropathic pain. Pain Research and Treatment. 2014;2014:854560.
65. Paladini A et al. Palmitoylethanolamide in the Treatment of Failed Back Surgery Syndrome. Pain Research and Treatment. 2017;2017:1486010.
66. Bacci C et al. Randomized split-mouth study on postoperative effects of palmitoylethanolamide for impacted lower third molar surgery. ISRN Surgery. 2011;2011:917350.
67. Chirchiglia D et al. Nonsurgical lumbar radiculopathies treated with ultramicronized palmitoylethanolamide (umPEA): A series of 100 cases. Neurol Neurochir Pol. 2018 Jan-Feb;52(1):44-47.
68. Indraccolo U et al. Micronized palmitoylethanolamide/trans-polydatin treatment of endometriosis-related pain: a meta-analysis. Annali Dell'Istituto Superiore di Sanita. Apr-Jun 2017;53(2):125-134.
69. Cremon C et al. Randomised clinical trial: the analgesic properties of dietary supplementation with palmitoylethanolamide and polydatin in irritable bowel syndrome. Alimentary Pharmacology & Therapeutics. Apr 2017;45(7):909-922.
70. Kamboj SS et al. N-Acetylcysteine Inhibits Hyperglycemia-Induced Oxidative Stress and Apoptosis Markers in Diabetic Neuropathy. Journal of Neurochemistry, 2010; 112: 77-91.
71. Nakayama M et al. Suppression of N(epsilon)-(carboxymethyl)lysine generation by the antioxidant N-acetylcysteine. Peritoneal dialysis international. May-Jun 1999;19(3):207-210.
72. Selhub J et al. B vitamins, homocysteine, and neurocognitive function in the elderly. The American journal of clinical nutrition. Feb 2000;71(2):614s-620s.
73. Sanchez-Ramirez GM et al. Benfotiamine Relieves Inflammatory and Neuropathic Pain in rats. European Journal of Pharmacology, 2006; 530: 48-53.
74. Varkonyi T et al. Diabetic Neuropathy: New Strategies for Treatment. Diabetes, Obesity and Metabolism, 2008; 10: 99-108.
75. Fahmy E et al. Estimation of Serum Homocysteine Level in Patients with Type 2 Diabetic Neuropathy. Egyptian Journal of Neurology Psychiatry and Neurosurgery, 2010; 47(1): 59-66.
76. Mizukami H et al. Methylcobalamin Effects of Diabetic Neuropathy and Nerve Protein Kinase C in Rats. European Journal of Clinical Investigation, 2011; 41(4): 442-450.
77. Okada K et al. Methylcobalamin Increases Erk1/2 and Akt Activities through the Methylation Cycle and Promotes Nerve Regeneration in a Rat Sciatic Nerve Injury Model. Experimental Neurology, 2010;222: 191-203.
78. Jacobs AM et al. Management of Diabetic Small-Fiber neuropathy with Combination L-Methylfolate, Methylcobalamin, and Pyridocal 5’-Phosphate. Reviews of Neurological Diseases, 2011; 8(1/2): 1-9.
79. Fonseca VA et al. Metanx in Type 2 Diabetes with Peripheral Neuropathy: A Randomized Trial. The American Journal of Medicine (2013) 126, 141-149.
80. Wade RL et al. Impact of L-Methylfolate Combination therapy Among Diabetic Peripheral Neuropathy Patients. American Journal of Pharmacological Benefits, 2012; 4(5): 218-225.
81. Talaei A et al. Vitamin B12 may be more effective than nortriptyline in improving painful diabetic neuropathy. International Journal of Food Sciences and Nutrition, 2009;60 Suppl 5:71-6.
82. Dominguez JC et al. A prospective, open label, 24-week trial of methylcobalamin in the treatment of diabetic polyneuropathy. Journal of Diabetes Mellitus, 2012; 2(4): 408-412.
83. Scaglione F et al. Folate, folic acid and 5-methyltetrahydrofolate are not the same thing. Xenobiotica. 2014 May;44(5):480-8.
84. Bhatia P et al. Homocysteine excess: delineating the possible mechanism of neurotoxicity and depression. Fundam Clin Pharmacol. 2015 Dec;29(6):522-8.
85. Ambrosch A et al. Relation between homocysteinaemia and diabetic neuropathy in patients with Type 2 diabetes mellitus. Diabet Med. 2001 Mar;18(3):185-92.
86. Wang D et al. Serum folate, vitamin B12 levels and diabetic peripheral neuropathy in type 2 diabetes: A meta-analysis. Mol Cell Endocrinol. 2017 Mar 5;443:72-9.
87. Mottaghi T et al. Effect of folic acid supplementation on nerve conduction velocity in diabetic polyneuropathy patients. Neurol Res. 2019 Apr;41(4):364-8.
88. Sun J et al. Role of curcumin in the management of pathological pain. Phytomedicine. 2018 Sep 15;48:129-40.
89. Banafshe HR et al. Effect of curcumin on diabetic peripheral neuropathic pain: possible involvement of opioid system. Eur J Pharmacol. 2014 Jan 15;723:202-6.
90. Attia HN et al. Protective effects of combined therapy of gliclazide with curcumin in experimental diabetic neuropathy in rats. Behav Pharmacol. 2012 Apr;23(2): 153-61.
91. Li Y et al. Curcumin attenuates diabetic neuropathic pain by downregulating TNF-alpha in a rat model. Int J Med Sci. 2013;10(4):377-81.
92. Zhao WC et al. Curcumin ameliorated diabetic neuropathy partially by inhibition of NADPH oxidase mediating oxidative stress in the spinal cord. Neurosci Lett. 2014 Feb 7;560:81-5.
93. Meng B et al. Effects of curcumin on TTX-R sodium currents of dorsal root ganglion neurons in type 2 diabetic rats with diabetic neuropathic pain. Neurosci Lett. 2015 Sep 25;605: 59-64.
94. Meng B et al. Effects of curcumin on sodium currents of dorsal root ganglion neurons in type 2 diabetic neuropathic pain rats. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2015 Nov;31(6):541-8.
95. Nabavi SF et al. Curcumin: a natural product for diabetes and its complications. Curr Top Med Chem. 2015;15(23):2445-55.
96. Zhang DW et al. Curcumin and diabetes: a systematic review. Evid Based Complement Alternat Med. 2013;2013:636053
97. Meng B et al. Antioxidant and antiinflammatory activities of curcumin on diabetes mellitus and its complications. Curr Pharm Des. 2013;19(11):2101-13.
98. Gulati OP. Pycnogenol® in Metabolic Syndrome and Related Disorders. Phytother Res. 2015 Jul;29(7):949-68.
99. Spadea L et al. Treatment of Cascular Retinopathies with Pycnogenol. Phytotherapy Research, 2001; 15: 219-223.
100. Liu X et al. Antidiabetic effect of PycnogenolR French maritime pine bark extract in patients with diabetes type II. Life Sciences, 2004; 75: 2505-2513.
101. Schonlau F et al. Pycnogenol for Diabetic Retinopathy. International Ophthalmology, 2002; 24: 161-171.
102. Steigerwalt R et al. Pycnogenol® Improves Microcirculation, Retinal Edema, and Visual Acuity in Early Diabetic Retinopathy. Journal of Ocular Pharmacology and Therapeutics, 2009; 25(6): 537-540.
103. Zibadi S et al. Reduction of cardiovascular risk factors in subjects with type 2 diabetes by Pycnogenol supplementation. Nutr Res. 2008 May;28(5):315-20.
104. Mah E et al. Supplementation of a γ-tocopherol-rich mixture of tocopherols in healthy men protects against vascular endothelial dysfunction induced by postprandial hyperglycemia. J Nutr Biochem. 2013 Jan;24(1):196-203.
105. Masterjohn C et al. γ-Tocopherol abolishes postprandial increases in plasma methylglyoxal following an oral dose of glucose in healthy, college-aged men. J Nutr Biochem. 2012 Mar;23(3):292-8.
106. Pasinetti GM et al. Role of grape seed polyphenols in Alzheimer’s disease neuropathology. Nutr Diet Suppl. 2010 Aug 1; 2010(2): 97–103.
107. Feringa HHH et al. The effect of grape seed extract on cardiovascular risk markers: a meta-analysis of randomized controlled trials. J Am Diet Assoc. 2011 Aug;111(8):1173-81.
108. Kaur M et al. Anticancer and Cancer Chemopreventive Potential of Grape Seed Extract and Other Grape-Based Products. J Nutr. 2009 Sep; 139(9): 1806S–1812S.
109. Kar P et al. Effects of grape seed extract in Type 2 diabetic subjects at high cardiovascular risk: a double blind randomized placebo controlled trial examining metabolic markers, vascular tone, inflammation, oxidative stress and insulin sensitivity. Diabet Med. 2009 May;26(5):526-31.
110. Chiva Blanch G et al. Effects of red wine polyphenols and alcohol on glucose metabolism and the lipid profile: a randomized clinical trial. Clin Nutr. 2013 Apr;32(2):200-6.
111. Hokayem M et al. Grape polyphenols prevent fructose-induced oxidative stress and insulin resistance in first-degree relatives of type 2 diabetic patients. Diabetes Care. 2013 Jun;36(6):1454-61.
112. Littarru GP et al. Bioenergetic and antioxidant properties of coenzyme Q10: recent developments. Mol Biotechnol. 2007 Sep;37(1):31-7.
113. Amin MM et al. Novel CoQ10 antidiabetic mechanisms underlie its positive effect: modulation of insulin and adiponectine receptors, Tyrosine kinase, PI3K, glucose transporters, sRAGE and visfatin in insulin resistant/diabetic rats. PLoS One. 2014 Feb 20;9(2):e89169.
114. Kolahdouz Mohammadi R et al. The effect of coenzyme Q10 supplementation on metabolic status of type 2 diabetic patients. Minerva Gastroenterol Dietol. 2013 Jun;59(2):231-6.
115. Hodgson JM et al. Coenzyme Q10 improves blood pressure and glycaemic control: a controlled trial in subjects with type 2 diabetes. Eur J Clin Nutr. 2002 Nov;56(11):1137-42.
116. Hamilton SJ et al. Coenzyme Q10 improves endothelial dysfunction in statin-treated type 2 diabetic patients. Diabetes Care. 2009 May;32(5):810-2.
117. Lindberg S et al. Adiponectin, type 2 diabetes and cardiovascular risk. European Journal of Preventive Cardiology, 21 Nov 2013, 22(3):276-283
118. Yoritaka A et al. Randomized, double-blind, placebo-controlled pilot trial of reduced coenzyme Q10 for Parkinson's disease. Parkinsonism Relat Disord. 2015 Aug;21(8):911-6.
119. Zhang YP et al. Prophylactic and antinociceptive effects of coenzyme Q10 on diabetic neuropathic pain in a mouse model of type 1 diabetes. Anesthesiology. 2013 Apr;118(4):945-54.
120. Langsjoen PH et al. Supplemental ubiquinol in patients with advanced congestive heart failure. Biofactors. 2008;32(1-4):119-28.
121. Hosoe K et al. Study on safety and bioavailability of ubiquinol (Kaneka QH) after single and 4-week multiple oral administration to healthy volunteers. Regul Toxicol Pharmacol. 2007 Feb;47(1):19-28.
122. Alamdari A et al. An inverse association between serum vitamin D levels with the presence and severity of impaired nerve conduction velocity and large fiber peripheral neuropathy in diabetic subjects. Neurol Sci. 2015 Jul;36(7):1121-6.
123. Soderstrom LH et al. Association between vitamin D and diabetic neuropathy in a nationally representative sample: results from 2001-2004 NHANES. Diabet Med. 2012 Jan;29(1):50-5.
124. Esteghamati A et al. Non-linear contribution of serum vitamin D to symptomatic diabetic neuropathy: A case-control study. Diabetes Res Clin Pract. 2016 Jan;111:44-50.
125. Shillo P et al. Reduced vitamin D levels in painful diabetic peripheral neuropathy. Diabet Med. 2019 Jan;36(1):44-51.
126. Lips P et al. Vitamin D and type 2 diabetes. J Steroid Biochem Mol Biol. 2017 Oct;173:280-285.
127. Vondra K et al. Vitamin D and new insights into pathophysiology of type 2 diabetes. Horm Mol Biol Clin Investig. 2021 Mar 1;42(2):203-208.
128. Chatterjee R et al. Effect of intra-trial mean 25(OH)D level on diabetes risk, by race and weight: an ancillary analysis in the D2d randomized study cohort. Am J Clin Nutr. 2023 Jul;118(1):59-67.
129. Mohd Ghozali N et al. Mechanisms Linking Vitamin D Deficiency to Impaired Metabolism: An Overview. Int J Endocrinol. 2022 Jul 6;2022:6453882.
130. Ao T et al. The Effects of Vitamin D on Immune System and Inflammatory Diseases. Biomolecules. Nov 3 2021;11(11)
131. Ghadiri-Anari A et al. Dose vitamin D supplementations improve peripheral diabetic neuropathy? A before-after clinical trial. Diabetes Metab Syndr. 2019 Jan - Feb;13(1):890-3.
132. Shehab D et al. Prospective evaluation of the effect of short-term oral vitamin d supplementation on peripheral neuropathy in type 2 diabetes mellitus. Med Princ Pract. 2015;24(3):250-6.
133. Razzaghi R et al. The effects of vitamin D supplementation on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. J Diabetes Complications. 2017 Apr;31(4):766-72.
134. Yorek MA. Is Fish Oil a Potential Treatment for Diabetic Peripheral Neuropathy? Curr Diabetes Rev. 2018;14(4):339-49.
135. Yorek MA. The Potential Role of Fatty Acids in Treating Diabetic Neuropathy. Curr Diab Rep. 2018 Aug 25;18(10):86.
136. Gerbi A et al. Fish oil supplementation prevents diabetes-induced nerve conduction velocity and neuroanatomical changes in rats. J Nutr. 1999 Jan;129(1):207-13.
137. Li MY et al. Dietary fish oil inhibits mechanical allodynia and thermal hyperalgesia in diabetic rats by blocking nuclear factor-kappaB-mediated inflammatory pathways. J Nutr Biochem. 2015 Nov;26(11):1147-55.
138. Tatsumi Y et al. Omega-3 polyunsaturated fatty acids exert anti-oxidant effects through the nuclear factor (erythroid-derived 2)-related factor 2 pathway in immortalized mouse Schwann cells. J Diabetes Investig. 2019 May;10(3):602-12.
139. Soleimani Z et al. Clinical and metabolic response to flaxseed oil omega-3 fatty acids supplementation in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. J Diabetes Complications. 2017 Sep;31(9):1394-400.
140. Stirban A et al. Effects of n-3 fatty acids on macro- and microvascular function in subjects with type 2 diabetes mellitus. Am J Clin Nutr. 2010 Mar;91(3):808-13.
141. Kim H-J et al. Nicotinamide adenine dinucleotide: An essential factor in preserving hearing in cisplatin-induced ototoxicity. Hear Res. 2015 Aug;326:30-9.
142. Poulose N et al. Sirtuin regulation in aging and injury. Biochim Biophys Acta. 2015 Nov;1852(11):2442-55.
143. Braidy N et al. Age related changes in NAD+ metabolism oxidative stress and Sirt1 activity in wistar rats. PLoS One. 2011 Apr 26;6(4):e19194.
144. Gomes AP et al. Declining NAD(+) induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell. 2013 Dec 19;155(7):1624-38.
145. Yoshino J et al. Nicotinamide mononucleotide, a key NAD(+) intermediate, treats the pathophysiology of diet- and age-induced diabetes in mice. Cell Metab. 2011 Oct 5;14(4):528-36.
146. Canto C et al. NAD(+) Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab. 2015 Jul 7;22(1):31-53.
147. Imai S-I et al. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 2014 Aug;24(8):464-71.
148. Wen C et al. Taurine is Involved in Energy Metabolism in Muscles, Adipose Tissue, and the Liver. Mol Nutr Food Res. Jan 2019;63(2):e1800536.
149. Ito T et al. The potential usefulness of taurine on diabetes mellitus and its complications. Amino Acids. 2012 May;42(5):1529-39.
150. Franconi F et al. Taurine levels in plasma and platelets in insulin-dependent and non-insulin-dependent diabetes mellitus: correlation with platelet aggregation. Adv Exp Med Biol. 1994;359:419-24.
151. Sak D et al. The Relationship between Plasma Taurine Levels and Diabetic Complications in Patients with Type 2 Diabetes Mellitus. Biomolecules. 2019 Mar 11;9(3):96.
152. Tao X et al. The effects of taurine supplementation on diabetes mellitus in humans: A systematic review and meta-analysis. Food Chem (Oxf). Jul 30 2022;4:100106.
153. Masouleh SS et al. The Effects of TRX Suspension Training Combined with Taurine Supplementation on Body Composition, Glycemic and Lipid Markers in Women with Type 2 Diabetes. Nutrients. Nov 5 2021;13(11)
参考来源:
美国糖尿病协会
http://www.diabetes.org
美国梅奥诊所
www.mayoclinic.org
美国国立糖尿病、消化和肾病研究所
http://www.niddk.nih.gov
美国糖尿病资讯交换中心
http://diabetes.niddk.nih.gov
加拿大糖尿病协会
http://www.diabetes.ca
加拿大公众卫生局
http://www.phac-aspc.gc.ca
免责声明和安全信息
- 本信息(包括任何附带资料)不是为了取代医生或有关合格从业人士的建议或忠告。
- 任何人如果想要对本文涉及的药物、饮食、运动或其他生活方式的使用、或改变调整,以预防或治疗某一特定健康状况或疾病,应首先咨询医生或有关合格从业人士,并获得他/她们的许可。妊娠和哺乳妇女在使用本网站任何内容前,尤其应征求医生的意见。
- 除非另有说明,本网站所述内容仅适用于成人。
- 本网站所推荐的任何产品,消费者应该以实际的产品标签内容为准,尤其应关注重要的安全信息以及产品最新信息,包括剂量、使用方法和禁忌症等。
- 由于循证医学研究、文献及有关产品处于不断的变化中,本网站工作人员将尽力更新。
- 本网站不能保证所载文章内容、综合干预方案以及相关成分或产品述及的健康益处,也不承担任何责任。

